Key points
- Notify your health department if you suspect a patient may have Ebola disease.
- Do not delay testing for other more likely medical conditions when assessing for Ebola disease.
- It is essential you wear proper PPE and take infection control measures while assessing a patient with suspected Ebola disease.
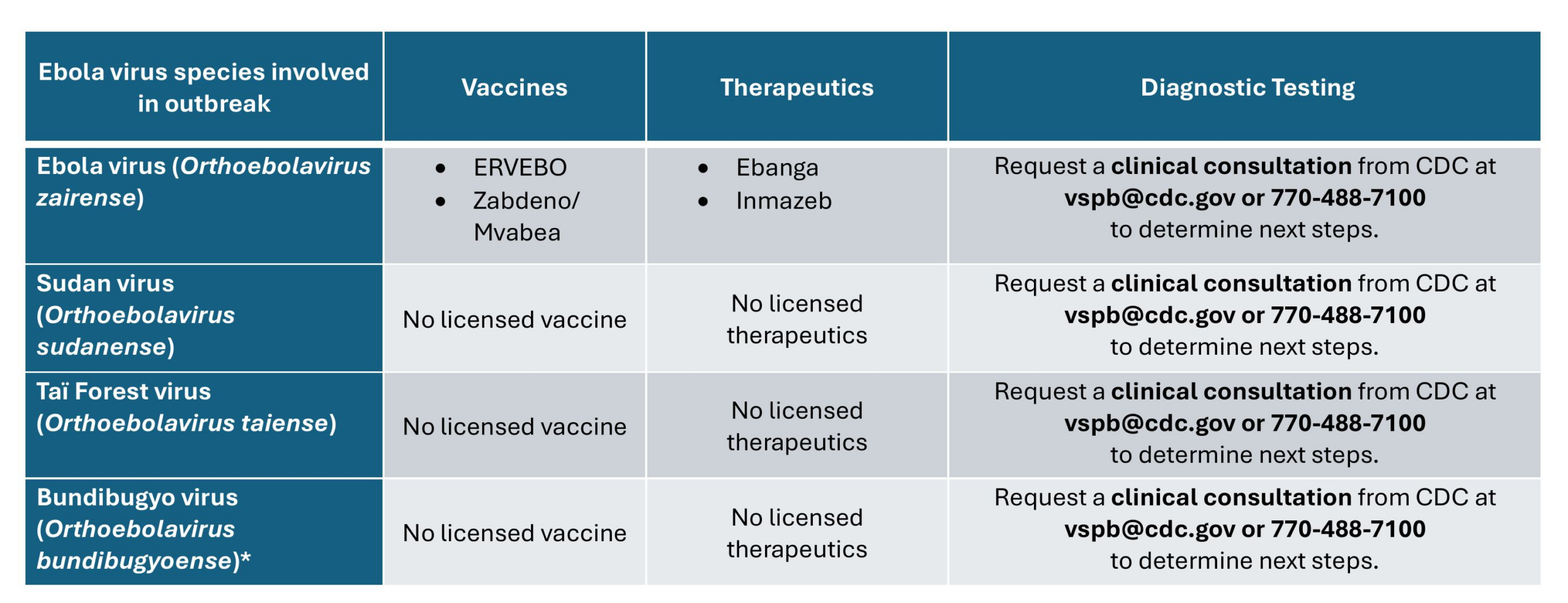
- FDA-approved treatments and vaccine are available only for Ebola disease caused by infection with Ebola virus (species Orthoebolavirus zairense).
- There are no FDA-approved treatments or vaccine for infection with the other three orthoebolaviruses that are known to cause disease in humans – Bundibugyo virus, Tai Forest virus, and Sudan virus
- People who receive early supportive care have better outcomes.
If you suspect Ebola disease in a patient
Assessments are critical
Most ill travelers returning from an area with an active Ebola disease outbreak or where Ebola disease is endemic are usually ill with a more common infection, such as malaria. Timely identification of other more likely pathogens and access to routine laboratory testing, such as blood counts and chemistries, is essential for providing appropriate patient care.
In the hospital setting, do not delay diagnostic assessments, laboratory testing, and appropriate care for other, more likely medical conditions while you're evaluating for Ebola disease. Use policies and procedures that are in place to safeguard healthcare workers.
Evaluation
Early consideration of Ebola disease in the differential diagnosis of a patient with consistent clinical and epidemiological factors is essential for providing appropriate and prompt patient care and preventing the spread of infection
Next steps if Ebola is suspected
Prevent transmission in healthcare settings
If a patient evaluation indicates possible infection with an orthoebolavirus, isolate the patient in a single room immediately, and adhere to infection prevention and control procedures to prevent transmission through direct or indirect contact, including wearing appropriate PPE and using dedicated equipment.
Notify your health department
Healthcare providers with concerns about Ebola infection during their patient's evaluation should contact their jurisdictional health department immediately (via the 24-hour Epi-On-Call contact list) and follow jurisdictional protocols for patient assessment.
Clinical teams should coordinate with public health officials and CDC to assess the risk of Ebola disease based on the clinical presentation and epidemiologic risk factors. This will help determine if testing is needed and what other causes of illness should be considered (e.g., malaria). This coordination can ensure proper patient care and appropriate precautions are taken to help prevent potential spread within the healthcare setting.
CDC's Viral Special Pathogens Branch (VSPB) is available 24/7 for consultations on Ebola disease by calling the CDC Emergency Operations Center at 770-488-7100 and requesting VSPB's on-call epidemiologist.
Treatment options
Therapeutics
Infection with orthoebolavirus zairense
Two treatments are approved by the U.S. Food and Drug Administration (FDA) to treat Ebola virus disease caused by Ebola virus (species Orthoebolavirus Zaire) .
Monoclonal antibodies, or mAbs, are artificially produced antibodies that act like natural antibodies to stop the virus from replicating in a person's body. These mAbs bind to a portion of the virus called the glycoprotein and prevent the virus from entering the person's cells.
- Inmazeb® is a combination of 3 monoclonal antibodies
- Ebanga® is a single monoclonal antibody.
Both treatments were evaluated in a randomized controlled trial in the Democratic Republic of the Congo. Overall survival was higher for patients who received either Inmazeb® or Ebanga®. The efficacy of these treatments has not been established for viruses other than Orthoebolavirus zairense.
Infection with other orthoebolaviruses that cause illness in humans
There are no Food and Drug Administration (FDA)-licensed or authorized vaccines or therapeutics for the other 3 orthoebolaviruses that cause human disease:
- Sudan virus, species Orthoebolavirus sudanense
- Taï Forest virus, species Orthoebolavirus taiense
- Bundibugyo virus, species Orthoebolavirus bundibugyoense
Potential therapeutic options for these viruses remain under investigation and are currently backed primarily by preclinical evidence.
*Certain antiviral and monoclonal antibody therapeutics are candidates for treatment but have limited efficacy data
Supportive care
Focus on supportive care for clinical management of patients with all forms of Ebola disease. This focus includes care for complications, which include hypovolemia, electrolyte abnormalities, hematologic abnormalities, refractory shock, hypoxia, hemorrhage, septic shock, multiorgan failure, and disseminated intravascular coagulation (DIC).
Recommended care includes:
- Volume repletion
- Blood pressure maintenance with vasopressors if needed
- Oxygenation maintenance
- Pain control
- Nutritional support
- Treatment of secondary bacterial infections and pre-existing comorbidities
- IV fluids, often in large volumes to correct dehydration due to diarrhea and vomiting. Some patients may develop profound third-spacing of fluids due to vascular leak.
Vaccine information
ERVEBO® is approved by the U.S. Food and Drug Administration (FDA) for the prevention of disease caused by Ebola virus (species Orthoebolavirus zairense) in individuals 12 months of age and older as a single dose administration. ERVEBO does not provide protection against other species of orthoebolaviruses or orthomarburgviruses.
Clinical guidance for specific populations
Pregnant women
Obstetric management of women who are pregnant with Ebola disease, particularly decisions about mode of delivery, needs to consider risks to the mother, risks of exposure for healthcare providers, and potential benefits to the neonate.
Neonates
Neonates born to patients with suspected or confirmed Ebola disease require special care and clinical safety precautions. Neonates born to patients with confirmed Ebola disease should not breastfeed. Decisions to discharge the neonate should be made in conjunction with local public health authorities.
Patients receiving hemodialysis
Follow protocols to safely perform renal replacement therapy (hemodialysis) in patients confirmed to have some VHFs including Ebola disease.
Ebola disease survivors
Ebola survivors can have many chronic complications, including viral persistence of orthoebolavirus in their bodies.
- Ansumana R, Jacobsen KH, Sahr F, Idris M, Bangura H, Boie-Jalloh M et al. Ebola in Freetown area, Sierra Leone—a case study of 581 patients. N Engl J Med. 2015 Feb 5;372(6):587–8.
- Bah EI, Lamah MC, Fletcher T, Jacob ST, Brett-Major DM, Sall AA et al. Clinical Presentation of Patients with Ebola Virus Disease in Conakry, Guinea. N Engl J Med. 2015 Jan 1;372(1):40–7.
- Baize S, Pannetier D, Oestereich L, Rieger T, Koivogui L, Magassouba N et al. Emergence of Zaire Ebola virus disease in Guinea. N Engl J Med. 2014 Oct 9;371(15):1418–25.
- Brett-Major DM, Jacob ST, Jacquerioz FA, Risi GF, Fischer WA 2nd, Kato Y et al. Beng Ready to Treat Ebola Virus Disease Patients. Am J Trop Med Hyg. 2015 Feb 4;92(2):233–7.
- Chertow DS, Kleine C, Edwards JK, Scaini R, Giuliani R, Sprecher A. Ebola Virus Disease in West Africa—Clinical Manifestations and Management. N Engl J Med. 2014 Nov 27;371(22):2054–7.
- Chertow DS, Nath A, Suffredini AF, Danner RL, Reich DS, Bishop RJ, Childs RW, Arai AE, Palmore TN, Lane HC, Fauci AS, Davey RT. Severe Meningoencephalitis in a Case of Ebola Virus Disease: A Case Report. Ann Intern Med. 2016 Apr 5. doi: 10.7326/M15–3066.
- Feldmann H, Geisbert TW. Ebola Haemorrhagic Fever. Lancet. 2011 Mar 5;377(9768):849–62.
- Fitzpatrick G, Vogt F, Moi Gbabai OB, Decroo T, Keane M, De Clerck H et al. The Contribution of Ebola Viral Load at Admission and Other Patient Characteristics to Mortality in a Médecins Sans Frontières Ebola Case Management Centre, Kailahun, Sierra Leone, June–October 2014. J Infect Dis. 2015 Dec 1;212(11):1752–8.
- Florescu DF, Kalil AC, Hewlett AL, Schuh AJ, Stroher U, Uyeki TM et al. Administration of Brincidofovir and Convalescent Plasma in a Patient With Ebola Virus Disease. Clin Infect Dis. 2015 Sep 15;61(6):969–73.
- Fowler RA, Fletcher T, Fischer WA 2nd, Lamontagne F, Jacob S, Brett-Major D et al. Caring for Critically Ill Patients with Ebola Virus Disease: Perspectives from West Africa. Am J Respir Crit Care Med. 2014 Oct 1;190(7):733–7.
- Hunt L, Gupta-Wright A, Simms V, Tamba F, Knott V, Tamba K et al. Clinical presentation, biochemical, and hematological parameters and their association with outcome in patients with Ebola virus disease: an observational cohort study. Lancet Infect Dis. 2015 Nov;15(11):1292–9.
- Jacobs M, Aarons E, Bhagani S, Buchanan R, Cropley I, Hopkins S et al. Post-exposure prophylaxis against Ebola virus disease with experimental antiviral agents: a case-series of health-care workers. Lancet Infect Dis. 2015 Nov;15(11):1300–4.
- Johnson DW, Sullivan JN, Piquette CA, Hewlett AL, Bailey KL, Smith PW et al. Lessons Learned: Critical Care Management of Patients With Ebola in the United States. Crit Care Med. 2015 Jun;43(6):1157–64.
- Kortepeter MG, Bausch DG, Bray M. Basic Clinical and Laboratory Features of Filoviral Hemorrhagic Fever. J Infect Dis. 2011 Nov;204 Suppl 3:S810–6.
- Kraft CS, Hewlett AL, Koepsell S, Winkler AM, Kratochvil CJ, Larson L, et al. The Use of TKM-100802 and Convalescent Plasma in 2 Patients With Ebola Virus Disease in the United States. Clin Infect Dis. 2015 Aug 15;61(4):496–502.
- Kreuels B, Wichmann D, Emmerich P, Schmidt-Chanasit J, de Heer G, Kluge S, et al. A Case of Severe Ebola Virus Infection Complicated by Gram-Negative Septicemia. N Engl J Med. 2014 Dec 18;371(25):2394–2401.
- Lai L, Davey R, Beck A, Xu Y, Suffredini AF, Palmore T wt al. Emergency Postexposure Vaccination With Vesicular Stomatitis Virus-Vectored Ebola Vaccine After Needlestick. JAMA. 2015 Mar 24–31;313(12):1249–55.
- Liddell AM, Davey RT Jr, Mehta AK, Varkey JB, Kraft CS, Tseggay GK et al. Characteristics and Clinical Management of a Cluster of 3 Patients With Ebola Virus Disease, Including the First Domestically Acquired Cases in the United States. Ann Intern Med. 2015 Jul 21;163(2):81–90.
- Lyon GM, Mehta AK, Varkey JB, Brantly K, Plyler L, McElroy AK et al. Clinical Care of Two Patients with Ebola Virus Disease in the United States. N Engl J Med. 2014 Dec 18;371(25):2402–2409.
- Mora-Rillo M, Arsuaga M, Ramírez-Olivencia G, de la Calle F, Borobia AM, Sánchez-Seco P et al. Acute respiratory distress syndrome after convalescent plasma use: treatment of a patient with Ebola virus disease contracted in Madrid, Spain. Lancet Respir Med. 2015 Jul;3(7):554–62.
- Parra JM, Salmerón OJ, Velasco M. The First Case of Ebola Virus Disease Acquired outside Africa. N Engl J Med. 2014 Dec 18;371(25):2439–40.
- Qin E, Bi J, Zhao M, Wang Y, Guo T, Yan T et al. Clinical Features of Patients With Ebola Virus Disease in Sierra Leone. Clin Infect Dis. 2015 Aug 15;61(4):491–5.
- Sagui E, Janvier F, Baize S, Foissaud V, Koulibaly F, Savini H et al. Severe Ebola Virus Infection With Encephalopathy: Evidence for Direct Virus Involvement. Clin Infect Dis. 2015 Nov 15;61(10):1627–8.
- Schibler M, Vetter P, Cherpillod P, Petty TJ, Cordey S, Vieille G et al. Clinical features and viral kinetics in a rapidly cured patient with Ebola virus disease: A case report. Lancet Infect Dis. 2015 Sep;15(9):1034–40.
- Schieffelin JS, Shaffer JG, Goba A, Gbakie M, Gire SK, Colubri A et al. Clinical Illness and Outcomes in Patients with Ebola in Sierra Leone. N Engl J Med. Nov 27;371(22):2092–100.
- Sueblinvong V, Johnson DW, Weinstein GL, Connor MJ Jr, Crozier I, Liddell AM et al. Critical Care for Multiple Organ Failure Secondary to Ebola Virus Disease in the United States. Crit Care Med. 2015 Oct;43(10):2066–75.
- Uyeki TM, Mehta AK, Davey RT Jr, Liddell AM, Wolf T, Vetter P et al. Clinical Management of Ebola Virus Disease in the United States and Europe. N Engl J Med. 2016 Feb 18;374(7):636–46.
- WHO Ebola Response Team, Agua-Agum J, Ariyarajah A, Blake IM, Cori A, Donnelly CA, Dorigatti I et al. Ebola virus disease among children in West Africa. N Engl J Med. 2015 Mar 26;372(13):1274–7.
- WHO Ebola Response Team. Ebola Virus Disease in West Africa—The First Nine Months of the Epidemic and Forward Projections. N Engl J Med. 2014 Oct 16;371(16):1481–95.
- WHO Ebola Response Team. West African Ebola Epidemic after One Year—Slowing but Not Yet under Control. N Engl J Med. 2015 Feb 5;372(6):584–7.
- ong KK, Davey RT Jr, Hewlett AL, Kraft CS, Mehta AK, Mulligan MJ, Beck A, Dorman W, Kratochvil CJ, Lai L, Palmore TN, Rogers S, Smith PW, Suffredini AF, Wolcott M, Ströher U, Uyeki TM. Use of post-exposure prophylaxis after occupational exposure to Zaire ebolavirus. Clin Infect Dis. 2016 Apr 26. pii: ciw256.
