Key points
- Patients with suspected mumps should be tested by rRT-PCR to confirm mumps at the beginning of a suspected mumps outbreak.
- Testing processes depend on symptoms and days since symptom onset.
- Contact your state or local health department to determine where to submit specimens and how to ship them.

When specimens should be collected
Optimal time to collect specimens
In an outbreak setting, the proportion of specimens with positive results by rRT-PCR (N target gene) decreased as the interval between onset and specimen collection increased1. The trend was reversed for IgM detection with increasing time to specimen collection the number of seropositive specimens increased.
The following graph shows the percentage of positive results obtained from testing 296 confirmed mumps cases from New York City by day of a sample collection after onset of symptoms. The results were determined positive by CDC IgM capture EIA or rRT-PCR (N target gene) as a function of time post parotitis (parotid gland swelling) onset.
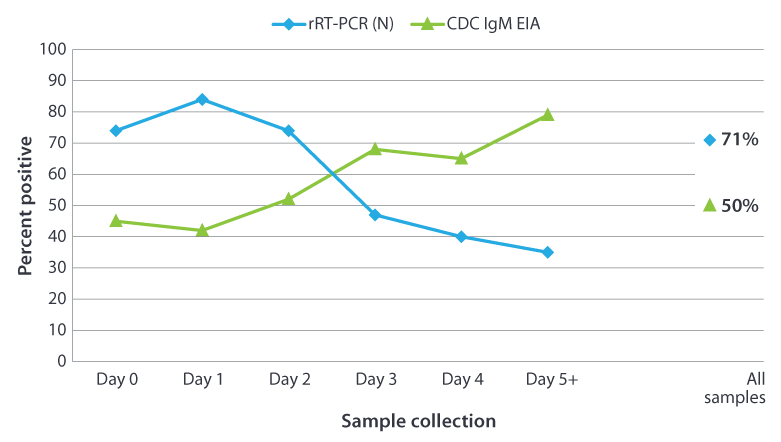
Notes about graph:
- The serum specimens were tested for presence of IgM using the CDC capture IgM EIA.
- The buccal swab specimens were tested by rRT-PCR using the mumps nucleoprotein (N) gene as the target.
- The mumps virus was isolated from 209 (71%) of the 296 buccal swabs tested.
- This testing was done in collaboration with New York City Department of Health and Mental Hygiene Public Health Laboratory, New York, New York.
Specimens to collect
CDC recommends the following for collecting specimens based on symptom onset. (For a visual tool, refer to Mumps Clinical Testing).
If ≤3 days since symptom onset
If it has been 3 or fewer days since symptom onset, collect a buccal swab specimen for detection of viral RNA by real-time (rRT-PCR). Collecting a buccal swab is important because:
- Buccal/oral swab specimen enhance the ability to laboratory-confirm a mumps infection.
- It can provide additional information (sequence analysis to obtain the mumps genotype) to aid epidemiologic investigations.
- It is necessary to obtain a buccal specimen to differentiate between an exposure to vaccine and an exposure to wild-type mumps virus. Serologic tests cannot differentiate between the two.
If >3 days since symptom onset
If it has been more than 3 days since symptom onset, collect a buccal swab specimen for rRT- PCR and a serum specimen for IgM detection. This may be particularly important in the beginning of an outbreak when it is unclear if mumps is the cause.
If patient presents certain signs
If the patient presents with any of the following, collect a buccal swab specimen for rRT-PCR and a serum specimen for IgM detection, regardless of days since symptom onset.
- Orchitis/oophoritis
- Mastitis
- Pancreatitis
- Hearing loss
- Meningitis
- Encephalitis
Specimen for serology
Collection instructions
CDC's Infectious Diseases Laboratories provide guidance for various specimen collection, storage, and shipment, including for mumps. Refer to the Infectious Diseases Laboratories Test Directory entry for each test or click the links below by test type. The instructions also include points of contact for the tests.
Instructions for test types
Watch video illustrating methods for collecting specimens:
Specimen submission
In addition to the information found on the CDC Infectious Diseases Laboratories Test Directory entry for each test, follow the guidance below for specimen collection and shipping. The instructions also include points of contact for the tests.
Contact health department first
Specimen shipping to CDC
If instructed to send specimens to CDC from within the Unites States, you must download and complete the CDC Specimen Submission Form (50.34). Please fill out a form for each specimen submitted. Submitters are encouraged to use the CDC Specimen Test Order and Reporting (CSTOR) Web Portal to submit specimens.
Provide the following information for shipments:
- Contact name with telephone number and email address
- Case ID and specimen ID from the submitting laboratory associated with the specimen
- Type of specimen, transport media (if applicable), and date collected
- MMR vaccination history [date(s), if known]
- Clinical signs and symptoms
- Patient's date of birth or age
- Onset date of parotitis or jaw swelling if applicable
- Any previous test results (if known)
- Rota JS, Rose JB, Doll MK, McNall RJ, McGrew M, Williams N, Lopareva EN, Barskey AE, Punsalang Jr A, Rota PA, Oleszko WR, Hickman CJ, Zimmerman DM, Bellini WJ. Comparison of the sensitivity of laboratory diagnostic methods from a well-characterized outbreak of mumps in New York City in 2009. Clin Vaccine Immunol. 2013;20:391-6.
- Hatchette TF, Mahony JB, Chong S, LeBlanc JJ. Difficulty with mumps diagnosis: what is the contribution of mumps mimickers? J Clin Virol 2009; 46:381-3.
- Jin L, Feng Y, Parry R, Cui A, Lu Y. Real-time PCR and its application to mumps rapid diagnosis. J Med Virol 2007; 79:1761-7.
- Krause CH, Eastick K, Ogilvie MM. Real-time PCR for mumps diagnosis on clinical specimens–comparison with results of conventional methods of virus detection and nested PCR. J Clin Virol 2006; 37:184-9.
- L'Huillier AG, Eshaghi A, Racey CS, et al. Laboratory testing and phylogenetic analysis during a mumps outbreak in Ontario, Canada. Virol J 2018; 15:98.
- Maillet M, Bouvat E, Robert N, et al. Mumps outbreak and laboratory diagnosis. J Clin Virol 2015; 62:14-9.
- Nunn A, Masud S, Krajden M, Naus M, Jassem AN. Diagnostic Yield of Laboratory Methods and Value of Viral Genotyping during an Outbreak of Mumps in a Partially Vaccinated Population in British Columbia, Canada. J Clin Microbiol 2018; 56.
- Tan KE, Anderson M, Krajden M, Petric M, Mak A, Naus M. Mumps virus detection during an outbreak in a highly unvaccinated population in British Columbia. Can J Public Health 2011; 102:47-50.
