At a glance
Introduction
Accurate and timely medical diagnoses support public health and patient care. In a 2015 report, "Improving Diagnosis in Healthcare," the National Academies of Medicine identified diagnostic error as a major public health challenge.1 Annually, approximately 800,000 Americans die or become disabled due to diagnostic errors associated with serious conditions that include cancer, cardiovascular, and infectious diseases.12 These diagnostic errors contribute to about one-third of all medical errors.
The Centers for Disease Control and Prevention (CDC) Division of Laboratory Systems (DLS) established the Diagnostic Excellence Initiative to incorporate laboratory expertise and capabilities into a diagnostic excellence enhancement strategy3.
The Total Testing Process
The Total Testing Process (TTP) in clinical settings is a step-by-step plan that guides everything from asking the right medical question and the correct test, to collecting the sample, analyzing it, and interpreting the results. By following these steps, healthcare teams support accurate diagnoses and promote patient safety.
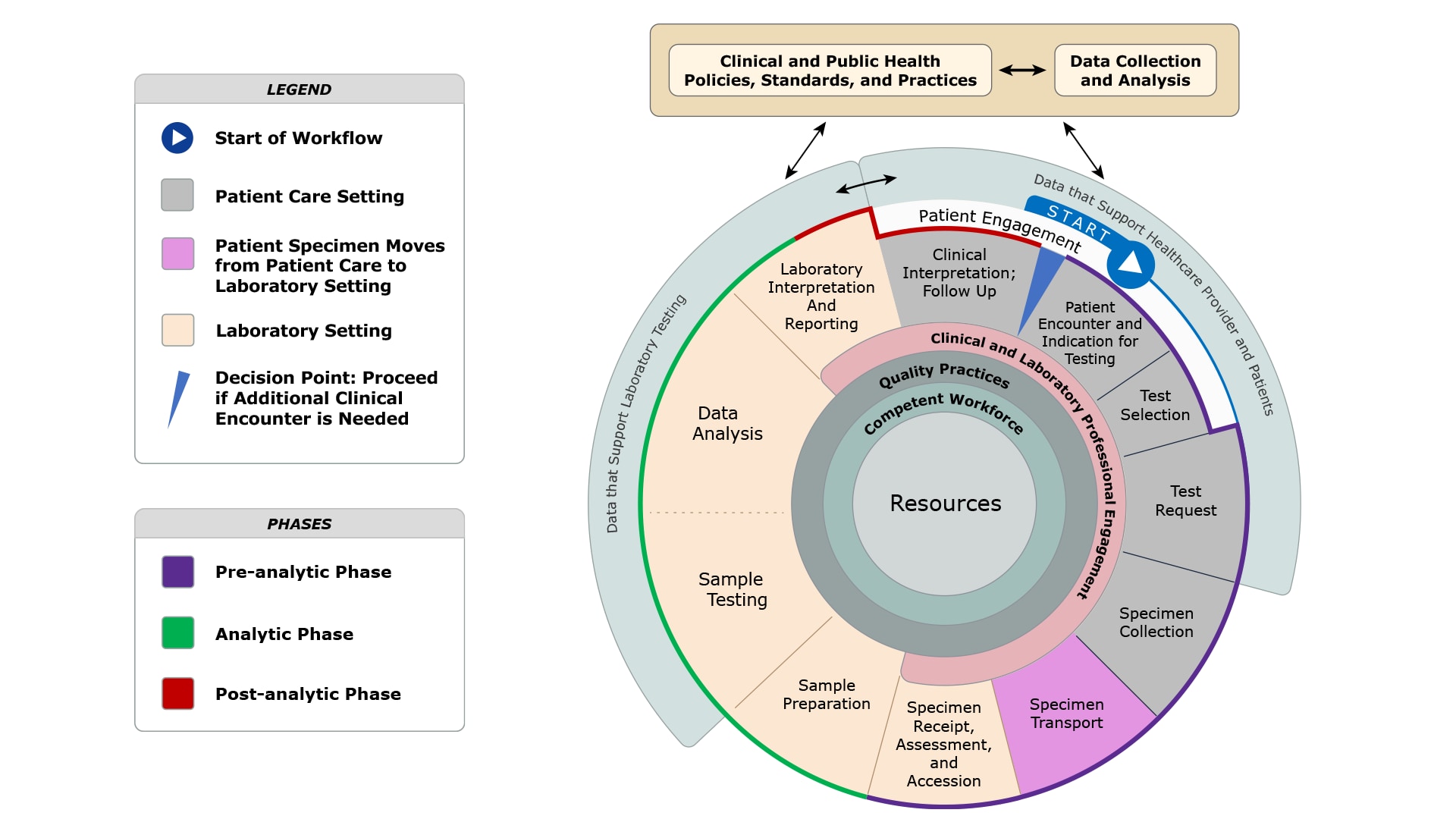
Phases of the Total Testing Process
- Pre-analytic Phase: Steps that occur prior to testing of the patient sample that begins with an indication for testing and concludes with specimen receipt, assessment, and accession.
- Analytic Phase: Steps that occur during testing of the patient sample that includes sample preparation, sample testing, and subsequent data analysis, laboratory interpretation and reporting.
- Post-analytic Phase: Steps that occur after testing of the patient sample that includes clinical interpretation and follow-up.
The following video demonstrates the TTP framework, using kidney function and disease testing as an example.
Key Areas Supporting Diagnostic Excellence
The Initiative advances diagnostic excellence through four closely linked focus areas:
- Enhancement of testing procedures and laboratory workflows to ensure accuracy and efficiency.
- Systematic evaluation of laboratory practices, fostering continuous improvement and adherence to high quality standards.
- Development of comprehensive policies, guidelines, and best practices to guide laboratory operations.
- Emphasis on educating patients and healthcare professionals, empowering them with the knowledge necessary to improve patient outcomes and support informed decision-making.
- The Clinical Laboratory Is an Integral Component to Health Care Delivery: An Expanded Representation of the Total Testing Process
- Prevalence, Awareness, and Treatment of Elevated LDL Cholesterol in US Adults
- Advancing Diagnostic Stewardship for Healthcare-Associated Infectious, Antibiotic Resistance, and Sepsis
- Practical Guidance for Clinical Microbiology Laboratories: A Comprehensive Update on the Problem of Blood Culture Contamination and a Discussion of Methods for Addressing the Problem
- National Kidney Foundation Laboratory Engagement Working Group Recommendations for Implementing the CKD-EPI 2021 Race-Free Equations for Estimated Glomerular Filtration Rate: Practical Guidance for Clinical Laboratories
- Reported Awareness and Adoption of 2021 Estimated Glomerular Filtration Rate Equations Among US Clinical Laboratories
