At a glance
In 2024, NHBS found 48% of persons who inject drugs (PWID) tested for HIV in the past year. Among HIV-negative PWID, 43% were aware of PrEP and 2% used it. Fifty-seven percent used syringe services programs, and 78% with HIV were taking antiretrovirals, indicating gaps in prevention and care.

Current data release
Read or download the current data release
NHBS PWID Data Release Figures, 2024
This PowerPoint slide set includes charts and figures for the 2024 NHBS Data Release
NHBS PWID Data Release Tables, 2024
Tables for the NHBS Data Release: HIV Infection Risk, Prevention, and Testing Behaviors Among Persons Who Inject Drugs: National HIV Behavioral Surveillance, 19 U.S. Cities, 2024
Key findings
Overview
This data release provides descriptive, unweighted data describing HIV infection, HIV testing, behavioral risk factors, and HIV prevention program participation among 9,237 persons who inject drugs (PWID) from the 2024 National HIV Behavioral Surveillance (NHBS). This release includes 19 U.S. cities in areas with high HIV prevalence. As a longstanding system for infectious disease behavioral surveillance, NHBS provides data that are used for tracking key indicators for populations of interest and to identify emerging trends in the HIV epidemic to inform real-world prevention strategies.
This summarizes the national data presented in this release. For complete results, refer to the slide set for figures and tables presenting national- and city-level data. See Technical Notes for information on definitions and data specifications.
NHBS topics
NHBS is the only source of national information on a comprehensive range of demographic, behavioral, and biological testing data on key populations, including PWID:
- Demographics
- Structural factors, including education, poverty, unemployment, health insurance, homelessness, and incarceration
- HIV prevalence
- HIV testing
- Sexual behaviors
- Injection drug use
- Injection equipment sharing behaviors
- Use of syringe services programs (SSPs) and pharmacies for sterile syringes
- Safe syringe disposal
- Pre-exposure prophylaxis (PrEP) awareness and use
- Sexually transmitted infections (STIs)
- Hepatitis C virus (HCV)
- Noninjection substance use
- Nonfatal opioid overdose
- Medications for opioid use disorder (MOUD)
- HIV care and treatment
Demographics
In 2024, 9,237 PWID participated in NHBS. Most PWID:
- were male (68%)
- were aged 50 years or older (37%), 40–49 years (28%), or 30–39 years (28%)
- were Black/African American (38%), White (34%), or Hispanic/Latino (20%)
- reported a disability (70%)
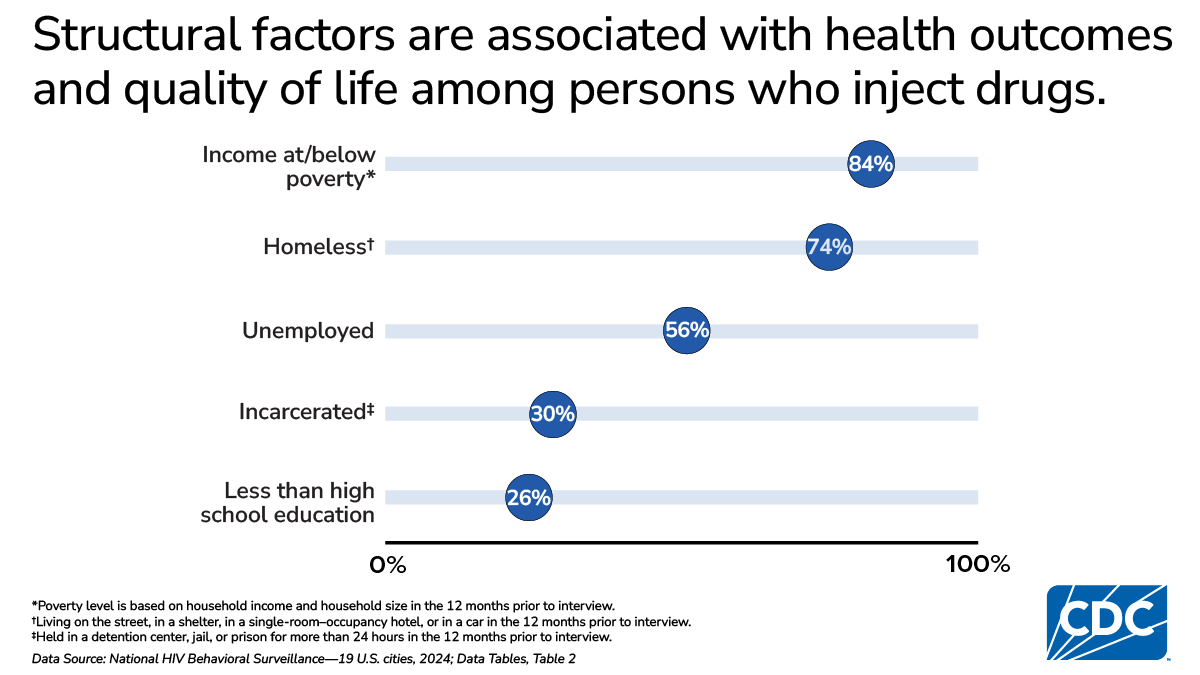
Structural factors
Structural factors refer to societal elements, including economic conditions, policies, laws, and social norms, that can influence health outcomes. Overall:
- 26% had less than a high school education
- 84% had income at or below the federal poverty level
- 56% were unemployed
- 74% were homeless in the past 12 months
- 30% were incarcerated in the past 12 months
- 13% had no health insurance
- 24% had not visited a health care provider in the past 12 months
HIV prevalence
One in 20 (5%) PWID with a valid NHBS HIV test result tested positive for HIV infection. HIV prevalence was the highest among:
- Males (6%)
- PWID aged 50 years or older (7%)
- Black/African American PWID (7%)
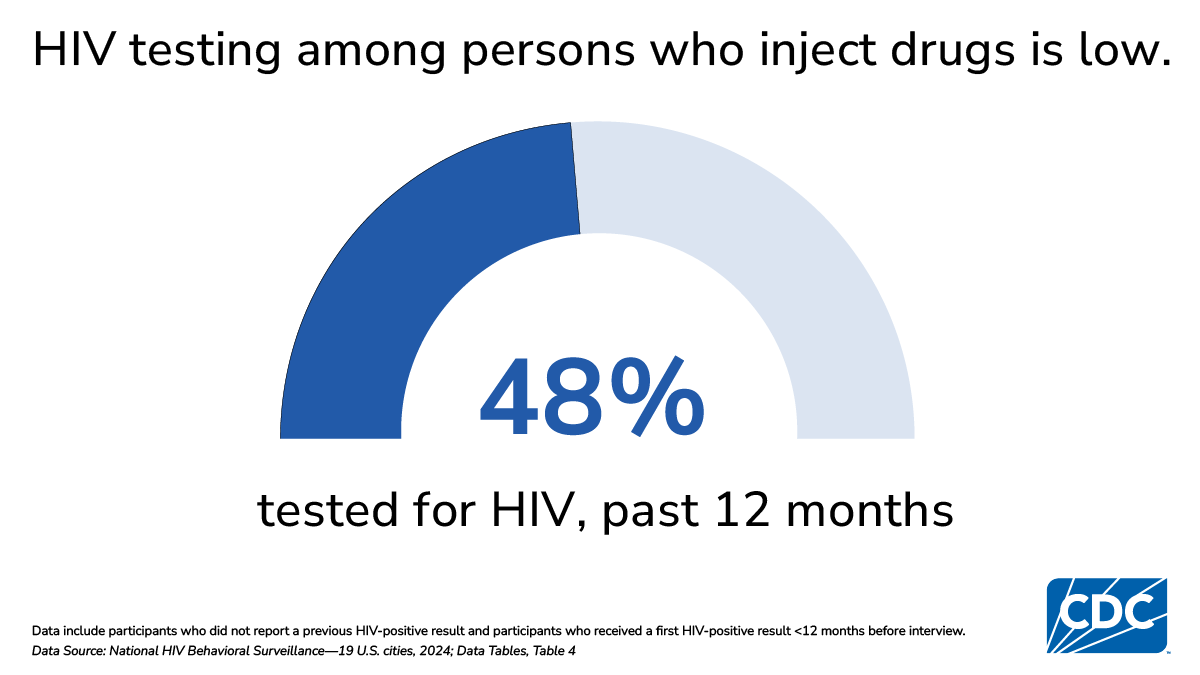
HIV testing
CDC recommends that persons at increased risk of HIV infection, including PWID, receive HIV testing at least annually.
- Among PWID who did not report a previous HIV-positive test result or who had received their first HIV-positive test result less than 12 months before the interview, 48% had an HIV test in the past 12 months
- Among PWID who had an HIV test in the past 12 months, 64% received their most recent test for HIV in a clinical setting
Sexual behaviors
Condomless sex
Condomless sex in the past 12 months was common among male and female PWID.
- 63% of male PWID reported condomless vaginal sex, 24% reported condomless anal sex with female sex partners, and 8% reported condomless anal sex with male sex partners
- 71% of female PWID reported condomless vaginal sex, and 26% reported condomless anal sex with male sex partners
- Among PWID with HIV, 27% of males and 57% of females reported condomless vaginal sex, and 14% of males and 23% of females reported condomless anal sex with a partner of the opposite sex
- 34% of males who tested HIV-positive reported condomless anal sex with male sex partners
Unprotected sex
"Unprotected sex" refers to sex without the participant's use of either condoms or HIV medications (i.e., HIV PrEP or antiretrovirals) with an HIV-discordant partner. "HIV discordant partner" refers to a sex partner of different or unknown HIV status.
- 29% of male PWID reported unprotected sex with an HIV-discordant partner at last sex
- 33% of female PWID reported unprotected sex with an HIV-discordant partner at last sex
Exchange sex
Giving or receiving money or drugs in exchange for sex is a recognized risk factor for HIV infection.
- 21% of male PWID reported giving money or drugs to a female in exchange for sex or giving or receiving money or drugs from a male in exchange for sex
- 29% of female PWID reported receiving money or drugs from a male in exchange for sex
Injection drug use
The prevalence of injection drug use by substance among PWID was as follows:
- Heroin: 74% past 12 months and 52% daily
- Fentanyl: 54% past 12 months and 36% daily
- Methamphetamine: 53% past 12 months and 27% daily
- Speedball: 48% past 12 months and 25% daily
- Powder or crack cocaine: 41% past 12 months and 18% daily
- Prescription opioids: 21% past 12 months and 7% daily
Injection equipment sharing behaviors
One-time use of sterile needles and syringes remains the safest, most effective way to limit HIV transmission during drug injection. Among PWID:
- 19% reported receptive sharing of syringes (using a syringe that had been used by someone else)
- 37% reporting receptive sharing of injection equipment
- 28% reported distributive sharing (giving a syringe they had already used to someone else); distributive syringe sharing was higher among PWID without HIV (28%) than PWID with HIV (17%)
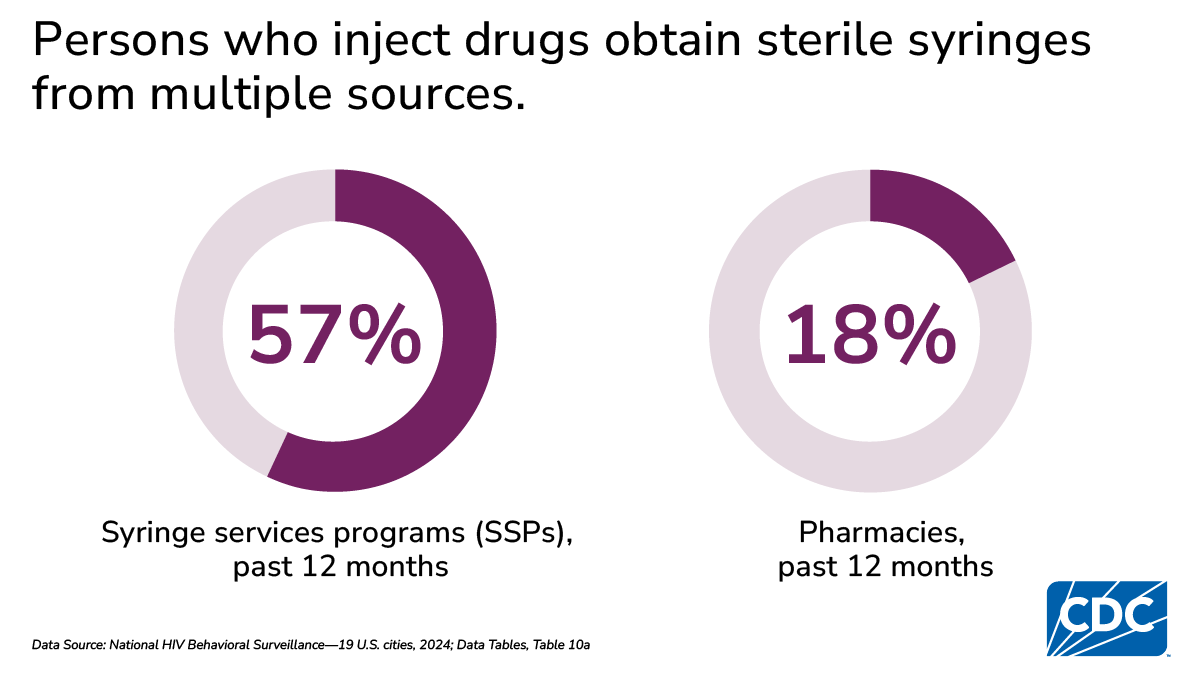
Use of syringe services programs (SSPs) and pharmacies for sterile syringes
SSPs are community-based prevention programs that can provide a range of services, including linkage to substance use treatment; access to, and disposal of, sterile syringes and injection equipment; and vaccination, testing, and linkage to care and treatment for infectious diseases. Receiving sterile syringes from SSPs and/or pharmacies reduces barriers to safer injection practices among PWID, which reduces infectious disease and increases access to other prevention services, including substance use treatment and overdose prevention.
- 57% of PWID reported receiving sterile syringes from SSPs, ranging by city from 3% to 85%
- 18% of PWID reported receiving sterile syringes from pharmacies, ranging by city from 6% to 43%
Safe syringe disposal
Access to safe syringe disposal can decrease the number of used syringes in the community and reduce accidental needle sticks.
- 27% of PWID reported disposing all their used syringes safely
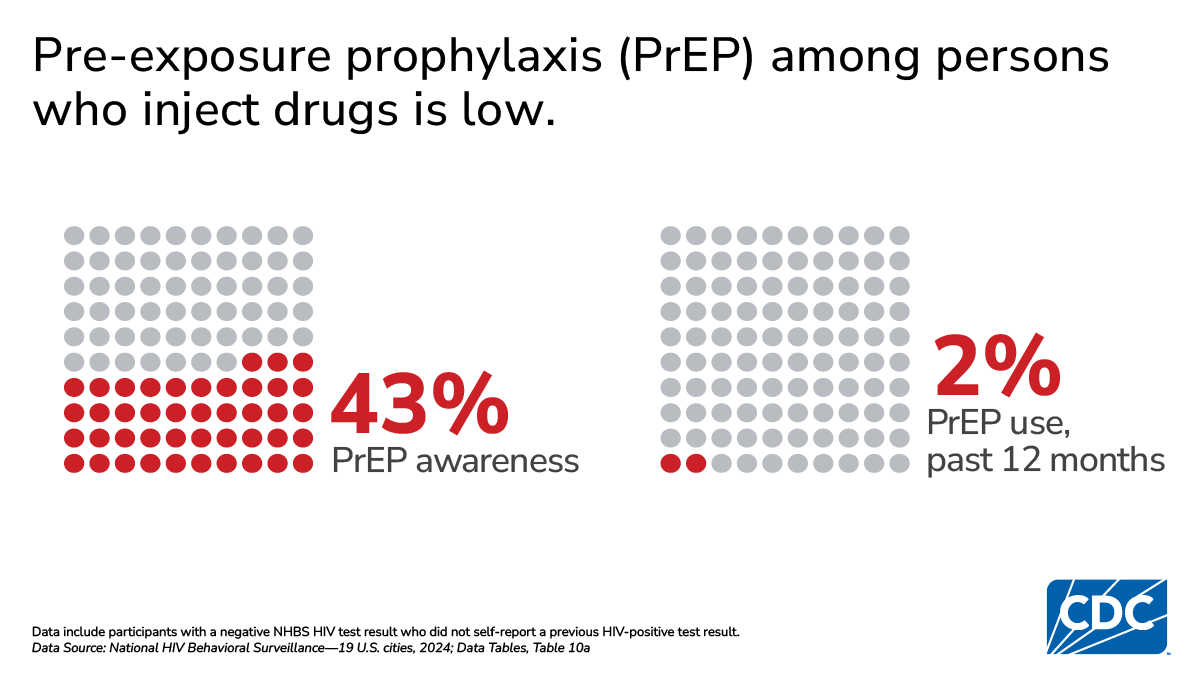
Pre-exposure prophylaxis (PrEP) awareness and use
CDC recommends the use of PrEP for persons at increased risk of HIV acquisition, including PWID.
- 43% of PWID without HIV were aware of PrEP, ranging by city from 10% to 68%
- 2% of PWID without HIV reported taking PrEP, ranging by city from 1% to 10%
Sexually transmitted infections (STIs)
STIs can increase the likelihood of acquiring and transmitting HIV.
- 8% of PWID reported a diagnosis of any bacterial STI (e.g., chlamydia, gonorrhea, or syphilis) in the past 12 months, including 22% of those with HIV and 8% of those without HIV
Hepatitis C virus (HCV)
HCV testing is recommended at least once for anyone who has ever injected drugs and annually for persons who currently inject drugs and who are thus at continued risk of infection.
- 75% of PWID reported ever testing for HCV
- 28% of PWID reported a previous diagnosis of hepatitis C, including 36% of those with HIV and 27% of those without HIV
- 14% of PWID reported having ever taken medicine to treat hepatitis C
Noninjection drug use
Noninjection drug use was common among PWID:
- 66% of PWID without HIV reported any noninjection drug use (excluding marijuana)
- 44% of PWID without HIV reported using methamphetamine, followed by heroin (41%) and crack (39%)
- 56% of PWID with HIV reported any noninjection drug use (excluding marijuana)
- 42% of PWID with HIV reported using methamphetamine, followed by crack (32%) and heroin (24%)
Nonfatal opioid overdose
Opioid overdose continues to be a major public health concern in the United States.
- 30% of PWID reported experiencing a nonfatal overdose in the past 12 months
Medications for opioid use disorder (MOUD)
Medications for opioid use disorder (MOUD), including methadone, buprenorphine, Suboxone, or Subutex, are effective, evidence-based treatment that decreases opioid use, opioid-related overdoses, and infectious disease transmission1.
- 50% of PWID who used opioids reported use of MOUD in the past 12 months
- 23% of PWID who used opioids tried but were unable to obtain MOUD in the past 12 months
- Among PWID without HIV, MOUD use (30%–75%) and unmet need for MOUD (15%-36%) varied by city
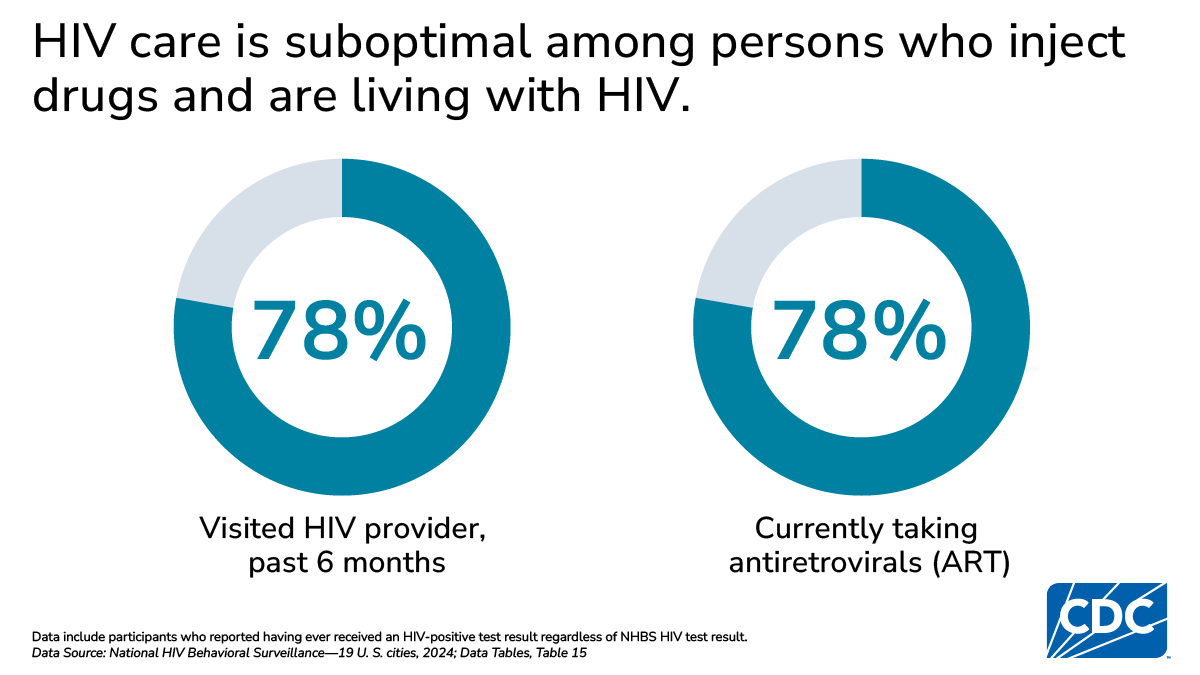
HIV care and treatment
CDC recommends that persons with HIV visit an HIV provider every 6 months and take antiretroviral treatment (ART). Taking ART as prescribed and keeping an undetectable viral load prevents HIV transmission and helps people with HIV stay healthy, live longer, and have better quality of life. Among PWID with HIV:
- 78% reported visiting a health care provider about HIV in the 6 months prior to interview
- 78% reported currently taking ART; 84% of Black/African American PWID, 76% of White PWID, and 69% of Hispanic or Latino PWID reported currently taking ART
Technical notes
National HIV Behavioral Surveillance (NHBS) conducts rotating cycles of biobehavioral surveys among men who have sex with men (MSM), persons who inject drugs (PWID), and heterosexually active persons at increased risk of HIV infection2; data are collected in annual cycles from 1 risk group per year, so that each population is surveyed once every 3 years. The same general eligibility criteria are used in each cycle: age 18 years or older, current residence in a participating city, no previous participation in NHBS during the current survey cycle, ability to complete the survey in either English or Spanish, and ability to provide informed consent. In addition to these basic NHBS eligibility criteria, participation in the 2024 NHBS cycle was limited to persons who reported injecting a drug that was not prescribed for them in the past 12 months.
A standardized questionnaire is used to collect information about behavioral risks for HIV infection, HIV testing, and use of HIV prevention services. The anonymous survey is administered in-person by a trained interviewer using a portable computer or tablet. All participants are offered an anonymous HIV test, which is linked to the survey data through a unique survey identifier.
Activities for NHBS are approved by CDC34 and by applicable institutional review boards in each participating city.
Participating cities
State and local health departments selected to participate in NHBS are among those whose jurisdictions include a metropolitan statistical area (MSA) or a specified division with the highest number of HIV infections reported to CDC during the 3-year time period 2017–2019. In 2024, NHBS was conducted in 19 MSAs (see list at the end of the report), which represented approximately 47% of all diagnoses reported to CDC in large MSAs (population ≥ 500,000) in the United States in 20225.
Throughout this report, MSAs and divisions are referred to by the name of the principal city.
Sampling method
The stigma associated with injection drug use presents challenges to sampling strategies for surveillance and research efforts among PWID6. Participants in the 2024 NHBS cycle were recruited using respondent-driven sampling789. Recruitment started with a limited number of initial participants ("seeds") who were chosen by referrals from people who knew the local population of PWID or through outreach to areas where PWID could be found. Initial participants who completed the eligibility screener and were eligible were administered the survey. Those who completed the survey were asked to recruit up to 5 PWID whom they knew personally and to give each recruit a coded coupon. Those recruits, in turn, completed the survey and were asked to recruit others. This recruitment process continued until the sample size was reached or the sampling period ended. Participants received incentives for participating in the survey and for recruiting others.
This activity was reviewed by CDC, deemed not research, and was conducted with applicable federal law and CDC policy
Data collection
Each person who brought a valid coupon to an NHBS field site was escorted to a private area with a trained interviewer for eligibility screening. If the person met eligibility requirements, the interviewer obtained informed consent and conducted the interview. The interview took approximately 40 minutes and consisted of questions about participants' demographic characteristics; HIV testing history; sexual and substance use behaviors; hepatitis C virus (HCV) testing, diagnosis, and treatment; sexually transmitted infection (STI) testing and diagnosis; and use of HIV prevention services and programs. In exchange for their time in the interview, participants received $25–$50 (amount determined locally).
For participants who consented to HIV testing, blood specimens were collected for rapid testing in the field with rapid or laboratory-based supplemental testing. HIV tests with a nonreactive rapid test result from participants who did not self-report a previous HIV-positive test result were considered HIV-negative; HIV tests with a reactive rapid test result were considered HIV-positive if supported by a second rapid test, supplemental laboratory-based test, or the participant's self-report of a previous HIV-positive test result. Participants received $20–$50 for HIV testing (amount determined locally).
Participants who agreed to recruit others received an additional incentive of $10–$25 for each recruit (up to 5) who completed the interview (amount determined locally). Each participating city's goal was to interview 500 PWID who reported injecting a drug that was not prescribed for them in the past 12 months.
Data analysis
This surveillance report presents descriptive data; no statistical tests were performed. In addition, these data are cross-sectional; we did not attempt to infer causal relationships. Reported numbers fewer than 12, and percentages based on these numbers, should be interpreted with caution because the numbers are considered unreliable.
Data for this report are not weighted. The purpose of this report is to provide a detailed summary of surveillance data collected as part of the NHBS 2024 cycle; unweighted data provide an efficient and transparent way to do so. Further, unweighted analysis allows for detailed reporting of outcomes among small subgroups of the population of interest.
In 2024, 19 MSAs participated in NHBS among PWID. Among the 19 MSAs, 11,139 persons were recruited to participate; 10,951 persons were screened to participate in NHBS. Of those, 1,519 did not meet NHBS eligibility criteria or did not provide consent and were excluded from the survey. An additional 195 interviews were excluded from this report due to incomplete survey data, survey responses of questionable validity, data lost during electronic upload, or missing data for sex.
The full analysis sample for this report includes 9,237 participants from the 2024 NHBS cycle. Additional inclusion criteria were applied for certain analyses of HIV infection, HIV-associated behaviors, and opioid use-related outcomes; details of each analysis sample can be found in the footnotes of each table.
Participating metropolitan statistical areas, 2024
Atlanta, Georgia
Atlanta–Sandy Springs–Alpharetta, Georgia
Baltimore–Columbia–Towson, Maryland
Chicago, Illinois
Chicago–Naperville–Elgin, Illinois–Indiana–Wisconsin (Chicago Division)
Denver, Colorado
Denver–Aurora–Lakewood, Colorado
Detroit, Michigan
Detroit–Warren–Dearborn, Michigan (Detroit Division)
Houston, Texas
Houston–The Woodlands–Sugar Land, Texas
Indianapolis, Indiana
Indianapolis-Carmel-Anderson, Indiana
Los Angeles, California
Los Angeles–Long Beach–Anaheim, California (Los Angeles Division)
New Orleans, Louisiana
New Orleans–Metairie, Louisiana
New York, New York
New York–Newark–Jersey City, New York–New Jersey–Pennsylvania (New York Division)
Newark, New Jersey
New York–Newark–Jersey City, New York–New Jersey–Pennsylvania (Newark Division)
Philadelphia, Pennsylvania
Philadelphia–Camden–Wilmington, Pennsylvania–New Jersey–Delaware–Maryland (Philadelphia Division)
Portland, Oregon
Portland–Vancouver–Hillsboro, Oregon–Washington
San Diego, California
San Diego–Chula Vista–Carlsbad, California
San Francisco, California
San Francisco–Oakland–Berkeley, California (San Francisco Division)
San Juan, Puerto Rico
San Juan–Bayamón–Caguas, Puerto Rico
Seattle, Washington
Seattle–Tacoma–Bellevue, Washington (Seattle Division)
Virginia Beach, VA
Virginia Beach–Norfolk–Newport News, Virginia–North Carolina
Washington, DC
Washington–Arlington–Alexandria, District of Columbia (DC)–Virginia–Maryland–West Virginia (Washington Division)
Definitions
Sociodemographic characteristics
- Sex: Male or female; n=2 intersex participants excluded from analysis by sex.
- Age: Calculated from the reported date of birth; age categories were chosen for epidemiologic relevance and consistency of reporting across all 3 National HIV Behavioral Surveillance (NHBS) populations.
- Race/ethnicity: Participants reported 1 or more race categories (American Indian or Alaska Native, Asian, Black or African American, Native Hawaiian or other Pacific Islander, and White). Hispanic or Latino ethnicity was asked separately; persons categorized by race did not report Hispanic or Latino ethnicity. The number of persons reported in each race category may, however, include persons whose ethnicity was not reported.
- Disability: Participants were classified as having a disability if they reported difficulty with hearing, seeing, cognition, ambulation, self-care, or independent living in response to the questions that comprise the U.S. Department of Health and Human Services data standard for disability status1011.
- City: Throughout this report, eligible metropolitan statistical areas (MSAs) and divisions are referred to by the name of the principal city. State and local health departments eligible to participate in NHBS are those with jurisdictions that include an MSA (or a specified division within an MSA) with high prevalence of HIV. This report presents 2024 data from 19 MSAs (see list at the end of the report).
Structural factors associated with health outcomes
- Less than high school education: Reported never attending school or having completed less than grade 12.
- Income at or below the federal poverty level: Participants were asked about their combined monthly or yearly household income (in US$) from all sources for the calendar year before interview. Poverty was determined by using the U.S. Department of Health and Human Services poverty guidelines for 2024. These guidelines are issued yearly for the United States and are one of the indicators used for determining eligibility for many federal and state programs. The 2024 guidelines12 were used for participants interviewed in 2024. Because the poverty guidelines are not defined for Puerto Rico, the guidelines for the 48 contiguous states and Washington, D.C. were used for this jurisdiction. Participants were asked to identify the range of their income by selecting from a list of income ranges and the number of dependents on that income. If the participant's income range and household size resulted in an ambiguous determination of poverty level, the participant's household income was assumed to be the low point of the income range.
- Unemployed: Participants who reported their employment status as "unemployed."
- Homeless: Living on the street, in a shelter, in a single-room–occupancy hotel, or in a car at any time in the 12 months before interview.
- Incarcerated: Having been held in a detention center, jail, or prison for more than 24 hours in the 12 months before interview.
- No current health insurance: Currently not having any form of health insurance.
- Did not visit a health care provider: Did not visit a healthcare provider in the 12 months before interview.
HIV status
HIV testing was performed for participants who consented to testing; blood specimens were collected for rapid testing in the field or supplemental laboratory-based testing.
- HIV-negative: Participants with a negative NHBS HIV test result who did not self-report a previous HIV-positive test result.
- HIV-positive: Participants who had a reactive rapid NHBS HIV test result that was supported by a second rapid test, supplemental laboratory-based testing, or self-report of a previous HIV-positive test result.
- No valid NHBS HIV test result: Participants who did not have a valid positive or negative NHBS HIV test result, including those who did not consent to the HIV test, had an indeterminate laboratory result, had discordant rapid test results, or reported a previous HIV-positive test result but had a negative NHBS HIV test result.
HIV testing
- Ever tested: Having had an HIV test during one's lifetime.
- Tested in past 12 months: Having had an HIV test in the 12 months before interview.
- Clinical setting: Participants reported the location of their most recent HIV test as private doctor's office (including health maintenance organization), emergency department, hospital (inpatient), public health clinic or community health center, family planning or obstetrics clinic, correctional facility (jail or prison), or drug treatment program.
- Nonclinical setting: Participants reported the location of their most recent HIV test as HIV counseling and testing site, HIV street outreach program or mobile unit, syringe services program, or home.
Sexual behaviors
- Anal sex: Penis inserted into a partner’s anus or butt.
- Vaginal sex: Penis inserted into a partner’s vagina.
- Oral sex: Mouth on a partner’s penis or vagina
- Condomless sex: Vaginal or anal sex during which a condom either is not used or is not used throughout the sex act.
- Number of sex partners: Median number of opposite sex partners during the 12 months before interview; first and third quartiles (25th and 75th percentiles) are also reported.
- Exchange sex for males: Refers to giving money or drugs to a female casual partner in exchange for sex or giving or receiving money or drugs from a male casual partner in exchange for sex.
- Exchange sex for females: Refers to receiving money or drugs from a male casual partner in exchange for sex.
- Casual partner: Person with whom the participant had sex, but to whom he or she does not feel committed or whom he or she does not know very well.
- Unprotected sex with HIV-discordant partner at last sex: “Unprotected sex” refers to sex without the participant’s use of either condoms or HIV medications (i.e., preexposure prophylaxis (PrEP) among those without HIV or antiretrovirals among those with HIV). “HIV-discordant partner” refers to a sex partner of different or unknown HIV status.
Injection drug use and behaviors
Participants were asked about their injection of specific drugs (excluding those prescribed for them) during the 12 months before interview.
- Years since first injection: Number of years since the participant first injected drugs not prescribed to them, based on the participant's reported age at first injection.
- Injected in the past 12 months: The participant reported injecting the specified drug at least once during the 12 months before interview.
- Injected daily: The participant reported injecting the specified drug daily during the 12 months before interview.
- Heroin: Injection of heroin.
- Fentanyl: Injection of fentanyl.
- Speedball: Injection of heroin and cocaine together through a single injection.
- Powder or crack cocaine: Injection of powdered cocaine or crack.
- Methamphetamine: Injection of methamphetamine.
- Prescription opioids: Injection of painkillers not prescribed for the participant, such as Oxycontin, Dilaudid, morphine, Percocet, or Demerol. The use of trade names is for identification only and does not imply endorsement by the Department of Health and Human Services or the Centers for Disease Control and Prevention.
- Other drug: Injection of any drug not prescribed for the participant, other than those listed above.
- Receptive sharing of syringes: Injecting with a syringe or needle that had already been used by someone else for injection.
- Receptive sharing of injection equipment: Using a cooker or cotton (used to filter particles from drug solution) that had already been used by someone else or using shared water for rinsing or injection.
- Receptive sharing of syringes to divide drugs: Dividing a drug solution by using a syringe that had already been used by someone else for injection.
- Any receptive sharing: Any combination of the 3 measures listed above (receptive sharing of syringes, receptive sharing of injection equipment, or receptive sharing of syringes to divide drugs).
- Distributive syringe sharing: A participant giving their syringe or needle to someone else to use after they had already used it for injection.
HIV prevention activities
- Receipt of syringes from syringe services programs (SSPs): Having received any new, sterile syringes from SSPs during the 12 months before interview.
- Receipt of syringes from pharmacy: Having received any new, sterile syringes from a pharmacy during the 12 months before interview.
- Substance use treatment: Participated in a substance use treatment program (including outpatient, inpatient, residential, detox, or 12-step program) during the 12 months before interview.
- Safe syringe disposal only: Disposed of syringes by putting them in a medical waste container or by exchanging them at an SSP, and no unknown or unsafe disposal method was indicated during the 12 months before interview.
- PrEP awareness: Ever heard of pre-exposure prophylaxis (PrEP), an antiretroviral medicine taken for months or years by a person who is HIV-negative to reduce the risk of getting HIV.
- PrEP use: Took PrEP at any point during the 12 months before interview to reduce the risk of getting HIV.
- Free condoms: Having received free condoms during the 12 months before interview, not including those given by a friend, relative, or sex partner.
Sexually transmitted infections (STIs)
- Any bacterial STI diagnosis: During the 12 months before interview, having been told that he or she had chlamydia, gonorrhea, or syphilis by a doctor or other health care provider.
- Chlamydia diagnosis: During the 12 months before interview, having been told that he or she had chlamydia by a doctor or other health care provider.
- Gonorrhea diagnosis: During the 12 months before interview, having been told that he or she had gonorrhea by a doctor or other health care provider.
- Syphilis diagnosis: During the 12 months before interview, having been told that he or she had syphilis by a doctor or other health care provider.
- Genital warts diagnosis: Having ever been told that he or she had genital warts by a doctor or other health care provider.
- Genital herpes diagnosis: Having ever been told that he or she had genital herpes by a doctor or other health care provider.
Hepatitis C virus (HCV) infection
- Hepatitis C testing: Having ever had a hepatitis C test.
- Hepatitis C diagnosis: Having ever been told that he or she had hepatitis C by a doctor or other health care provider.
- Hepatitis C treatment: Having ever taken medicine to treat hepatitis C.
Noninjection drug use
Participants were asked about their use of drugs (excluding those prescribed for them) during the 12 months before interview and their use of alcohol during the 30 days before interview. Participants were not limited in the number of substances they could report. Participants were considered to have used a substance if they reported using that substance with any frequency other than "never." The use of trade names is for identification only and does not imply endorsement by the Department of Health and Human Services or the Centers for Disease Control and Prevention.
- Any noninjection drug: Used any noninjection drug, excluding marijuana, during the 12 months before interview.
- Binge Drinking: Consumed 5 or more alcoholic drinks (males) or 4 or more alcoholic drinks (females) in about 2 hours during the 30 days before interview.
- Cocaine: Used powder cocaine during the 12 months before interview.
- Crack: Used crack cocaine during the 12 months before interview.
- Downer: Used downers (benzodiazepines), such as Klonopin, Valium, Ativan, or Xanax, during the 12 months before interview.
- Ecstasy: Used X or ecstasy during the 12 months before interview.
- Heroin: Used heroin (smoked or snorted) during the 12 months before interview.
- Marijuana: Used marijuana during the 12 months before interview.
- Methamphetamine: Used methamphetamines, including meth, crystal meth, speed, or crank, during the 12 months before interview.
- Prescription opioids: Used painkillers, such as OxyContin, Vicodin, morphine, or Percocet, during the 12 months before interview.
Opioid use-related outcomes
Opioid use-related outcomes were assessed for participants who reported injection or noninjection use of heroin or other opioids not prescribed for them during the 12 months before interview. The use of trade names is for identification only and does not imply endorsement by the Department of Health and Human Services or the Centers for Disease Control and Prevention.
- Used medications for opioid use disorder (MOUD): Having taken medicines like methadone, buprenorphine, Suboxone, or Subutex to treat drug use during the 12 months before interview.
- Unmet need for MOUD: Participant reported trying but being unable to obtain medicines, such as methadone, buprenorphine, Suboxone, or Subutex, to treat drug use during the 12 months before interview.
- Nonfatal opioid overdose: Having passed out, turned blue, or stopped breathing from using heroin or painkillers during the 12 months before interview.
Receipt of HIV care
Participants who reported having previously received a positive HIV test result were asked about their receipt of HIV care.
- Visited health care provider about HIV, ever: Having ever visited a health care provider for HIV care.
- Visited health care provider about HIV, within 1 month after diagnosis: Having visited a health care provider for HIV care within 1 month of the date of their first HIV-positive test result.
- Visited health care provider about HIV, in the past 6 months: Having visited a health care provider for HIV care during the 6 months before date of interview.
- Currently taking antiretroviral HIV medicines: Taking antiretroviral medicines at the time of interview.
Using these data
Acknowledgements
Atlanta, GA: Genetha Mustaafaa, Jenna Gettings, Christian Nelson; Baltimore, MD: Colin Flynn, Danielle German, Molly Gribbin; Chicago, IL: Antonio D. Jimenez, Irina Tabidze; Denver, CO: Alia Al-Tayyib, Jessica Forsyth, Megan Duffy; Detroit, MI: Emily Higgins, Vivian Griffin; Houston, TX: Salma Khuwaja, Paige Padgett, Osaro Mgbere; Indianapolis, IN: Daniel Hillman, Conner Tiffany, Manuel Gonzalez-Carrasco; Los Angeles, CA: Ekow Kwa Sey, Yingbo Ma; New Orleans, LA: William T. Robinson, Meredith Booth; New York City, NY: Abdier Benitez Flete, Alexis Rivera, Kristina Rodriguez; Newark, NJ: Afework Wogayehu, Anindita Fahad, Corey Rosmarin-DeStafano; Philadelphia, PA: Kathleen A. Brady, Tanner Nassau; Portland, OR: Lalaine Sevillano, Matthew Town, Lauren Lipira; San Diego, CA: Kristin Akerele, Stephanie Sanz, Nabeeh Hasan; San Francisco, CA: Erin C Wilson, Joaquin Meza, Willi McFarland; San Juan, PR: Yadira Rolón-Colón, Jesus Vargas-Franco, María Pabón Martínez; Seattle, WA: Sara Glick, Aleks Martin, Courtney Moreno; Virginia Beach, VA: Jennifer Kienzle, Jamell James, Gregg Fordham; Washington, DC: Irene Kuo, Kate Drezner, Sydney Bornstein; CDC: Amy Baugher, Julie Berg, Dita Broz, Janet Burnett, Susan Cha, Johanna Chapin-Bardales, Jacklynn De Leon, Paul Denning, Patrick Eustaquio, Lyssa Faucher, Jonathan Feelemyer, Teresa Finlayson, Savannah Harris, Maya Haynes, Rebecca Hershow, Dafna Kanny, Kathryn Lee, Rashunda Lewis, Danielle Lonbong Njiometio, Elana Morris, Evelyn Olansky, Adeolu Orogade, Catlainn Sionean, Larshie Sutter, Anna Teplinskaya, Jeffery Todd, Cyprian Wejnert.
Suggested citation
All material contained in this report is the public domain and may be used and reprinted without special permission; however, citation as to source is appreciated.
HIV Risk, Prevention, and Testing Behaviors Among People Who Inject Drugs—National HIV Behavioral Surveillance, 19 U.S. Cities, 2024. Centers for Disease Control and Prevention. Published [date]. Accessed [date]. https://www.cdc.gov/hiv-data/nhbs/hiv-risk-prevention-testing-pwid.html
- MacArthur GJ, Minozzi S, Martin N, et al. Opiate substitution treatment and HIV transmission in people who inject drugs: systematic review and meta-analysis. BMJ. 2012-10-04 23:32:25 2012;345. doi:10.1136/bmj.e5945.
- Kanny D, Broz D, Finlayson T, Lee K, Sionean C, Wejnert C; NHBS Study Group. A key comprehensive system for biobehavioral surveillance of populations disproportionately affected by HIV (National HIV Behavioral Surveillance): Cross-sectional survey study. JMIR Public Health Surveill 2022;8(11):e39053. doi:10.2196/39053.
- CDC. Guidelines for Defining Public Health Research and Public Health Non-Research. https://www.cdc.gov/scientific-integrity/media/pdfs/2024/10/defining-public-health-research-non-research-1999.pdf. Published October 4, 1999. Accessed July 18, 2025.
- Protection of Human Subjects, CFR 45, Part 46. https://www.ecfr.gov/current/title-45/subtitle-A/subchapter-A/part-46. Last amended January 12, 2023. Accessed July 18, 2025.
- CDC. Diagnoses, deaths, and prevalence of HIV in the United States and 6 territories and freely associated states, 2022. HIV Surveillance Report, 2022; vol. 35. https://stacks.cdc.gov/view/cdc/156509. Published May 2024. Accessed July 18, 2025.
- Watters J, Biernacki P. Target Sampling: Options for the Study of Hidden Populations. Soc Probl. 1989;36(4):416-430. doi:10.2307/800824.
- Heckathorn DD. Respondent-driven sampling: A new approach to the study of hidden populations. Soc Probl 1997;44(2):174-99. doi:10.2307/3096941.
- Heckathorn DD. Respondent-driven sampling II: Deriving valid population estimates from chain-referral samples of hidden populations. Soc Probl. 2002;49(1):11-34. doi:10.1525/sp.2002.49.1.11.
- Lansky A, Abdul-Quader AS, Cribbin M, et al. Developing an HIV behavioral surveillance system for injecting drug users: the National HIV Behavioral Surveillance System. Public Health Rep 2007;122(suppl 1):48–55. doi:10.1177/ 00333549071220S108.
- U.S. Census Bureau [Brault M, Stern S, Raglin D]. Evaluation report covering disability. https://www.census.gov/content/dam/Census/library/working-papers/2007/acs/2007_Brault_01.pdf. Published January 2007. Accessed July 18, 2025.
- Office of Minority Health. Data collection standards for race, ethnicity, sex, primary language, and disability status. https://aspe.hhs.gov/reports/hhs-implementation-guidance-data-collection-standards-race-ethnicity-sex-primary-language-disability. Published October 2011. Accessed July 18, 2025.
- U.S. Department of Health and Human Services. Prior HHS poverty guidelines and Federal Register references. https://aspe.hhs.gov/topics/poverty-economic-mobility/poverty-guidelines/prior-hhs-poverty-guidelines-federal-register-references. Accessed July 18, 20
