At a glance
CDC's newborn screening (NBS) laboratories provide states with quality control dried blood spot materials, technical assistance, and training. This is how CDC assures that screenings to identify serious but treatable conditions remain reliable for millions of babies each year.
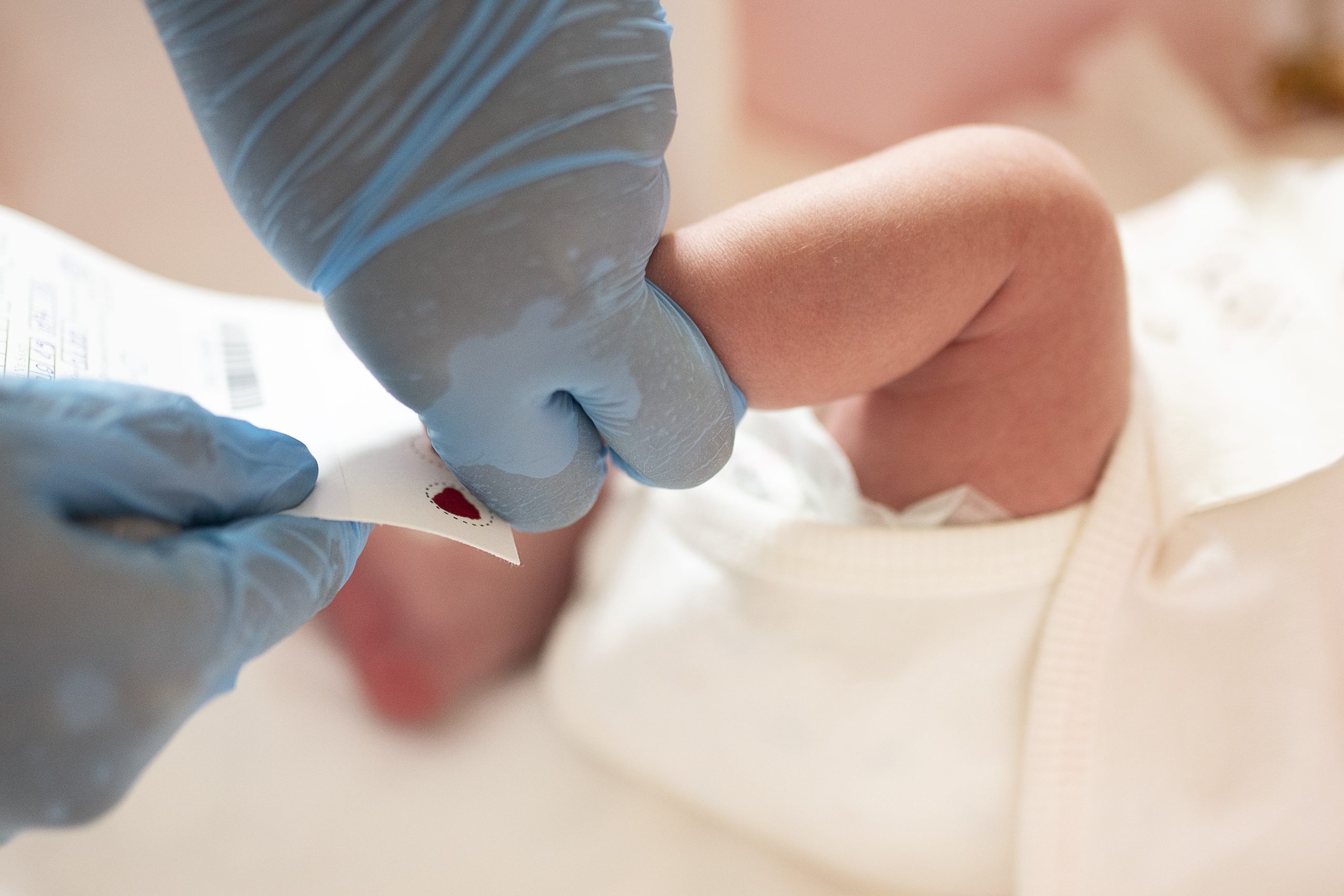
How does CDC support NBS testing quality?
Through the Newborn Screening and Quality Assurance Program (NSQAP), CDC provides resources and services to state NBS programs to ensure timely and accurate screening, which is essential for early identification of many serious conditions that affect thousands of babies born each year.
As part of NSQAP, CDC staff create dried blood spot quality assurance (QA) materials that closely mimic these conditions. State laboratories then use these materials to check their testing methods and results. This process helps ensure tests are accurate so that affected babies can be diagnosed and treated as early as possible.
Does CDC develop NBS tests and methods?
Yes. CDC develops new tests and enhances existing methods to improve the accuracy, reliability, and efficiency of NBS. CDC also works with public health laboratories to transfer and verify new methods and technologies.
Does CDC ever work with state labs directly?
Yes. CDC offers one-on-one support to troubleshoot and improve testing methods and share new technologies. CDC also conducts on-site evaluations to help states assess and improve their molecular testing processes. This support is part of the Molecular Assessment Program (MAP), developed in partnership with the Association of Public Health Laboratories (APHL).
To request remote or in-person technical assistance, please contact NSQAPDMT@cdc.gov.
Does CDC train state laboratory staff in NBS methods?
Yes. CDC hosts hands-on workshops to help state lab personnel learn about and master both molecular and biochemical mass spectrometry testing methods. These workshops equip state laboratory staff with the skills they need to effectively use the latest NBS techniques.
Calls for workshop applications are managed by the APHL Newborn Screening & Genetics Program.
How does CDC make sure its products are reliable and accurate?
The CDC’s Newborn Screening and Molecular Biology Branch (NSMBB) is committed to high-quality practices in NBS laboratories. NSMBB has achieved ISO/IEC 17034 and 17043 accreditations from the American Association for Laboratory Accreditation (A2LA). This accreditation means that the NSMBB laboratory has been reviewed by an outside group to ensure it meets recognized standards, adding an important level of quality control. Accreditation helps ensure that states can accurately and quickly detect serious conditions in babies—leading to better health outcomes and greater peace of mind for families.
Contacts
Please note that CDC cannot respond to questions about individual medical cases, provide second opinions, or make specific recommendations regarding therapy. Those issues should be addressed directly with your health care provider.
E-mail: CDC-INFO
Phone: CDC Health Line 1-800-CDC-INFO (1-800-232-4636)
Address:
Division of Laboratory Sciences
Mail Stop F-204770 Buford Highway
NE Atlanta, GA 30341-3724
