At a glance
CDC engages with the laboratory and testing community, provides reliable and timely laboratory testing during infectious disease outbreaks and harmful exposures, and prioritizes high-quality laboratory science so that we are ready to respond to health threats, anytime.

Overview

Recent infectious disease outbreaks and public health emergencies highlighted the importance of laboratory diagnostic testing as a core capability of the U.S. public health system—especially at the beginning of emergency responses when there is a greater demand for laboratory testing.
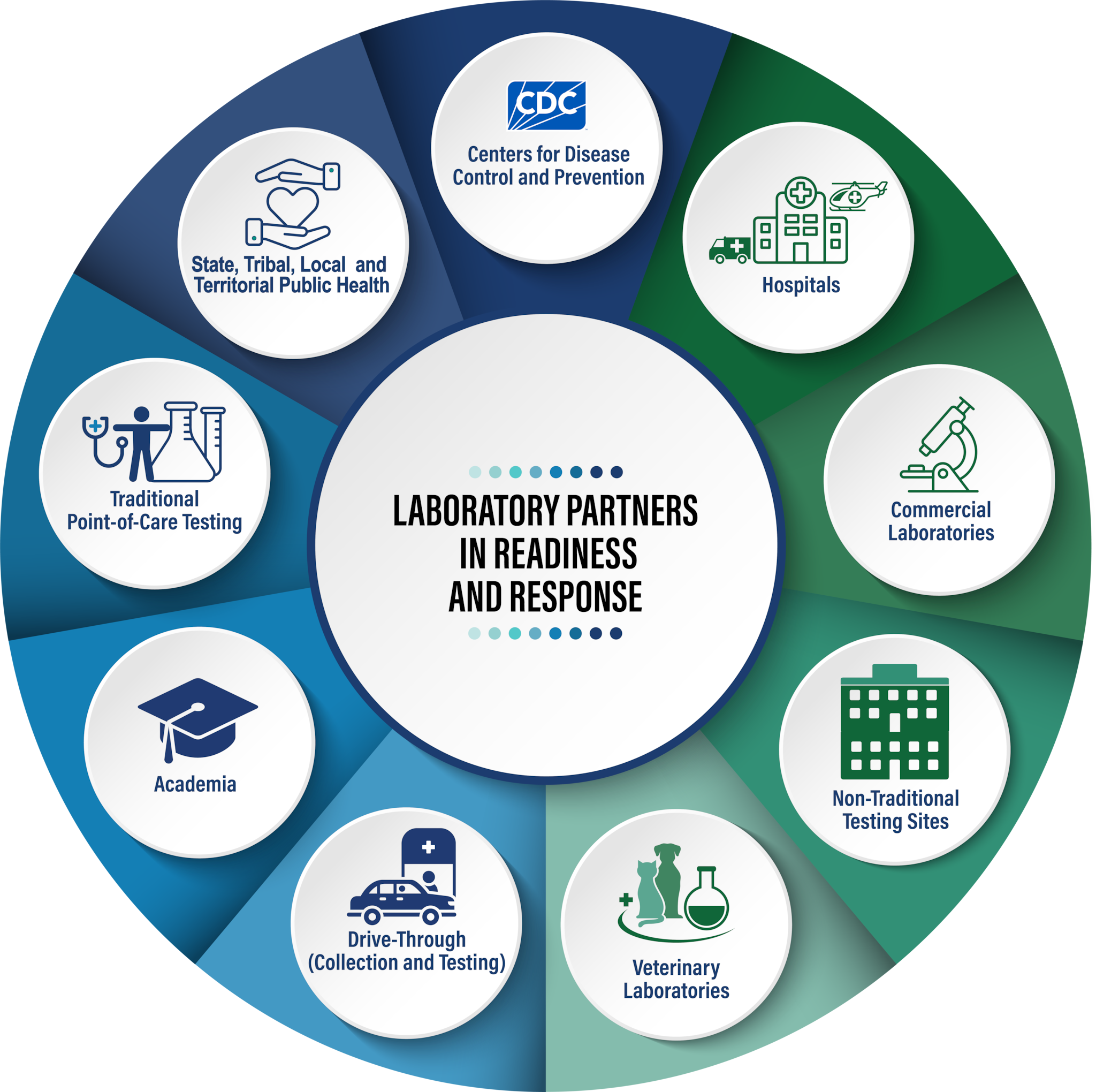
CDC demonstrates its commitment to laboratories by elevating and aligning laboratory support systems to focus on these key priorities:
Reshaping the future of laboratory readiness and response
- Interagency coordination and cross-cutting laboratory operation and systems support within CDC and beyond
Engagement with public and private-sector partners including the nation's clinical and public health laboratory systems and networks
- Memorandum of Understanding (MOU) for Diagnostic Surge Testing Capacity for Public Health Emergencies
- Clinical Laboratory Improvement Amendments of 1988 (CLIA)
- Laboratory Response Network (LRN)
