Cyanogen chloride (CK): Systemic Agent
CAS #:
506-77-4
RTECS #: GT2275000
UN #: 1589 (Guide 125)
Common Names:
- Chlorcyan
- Chlorine cyanide
Agent Characteristics
Colorless, liquid below 55°F (12.8°C) or gas above 55°F (12.8°C).
Cyanogen chloride (CK) is a highly volatile and toxic chemical asphyxiant that interferes with the body’s ability to use oxygen. Exposure to cyanogen chloride (CK) can be rapidly fatal. It has whole-body (systemic) effects, particularly affecting those organ systems most sensitive to low oxygen levels: the central nervous system (brain), the cardiovascular system (heart and blood vessels), and the pulmonary system (lungs). Cyanogen chloride (CK) has strong irritant and choking effects. Its vapors are extremely irritating and corrosive. Cyanogen chloride (CK) is a chemical warfare agent (military designation CK). It is used commercially in chemical synthesis and fumigation.
- Indoor Air: Cyanogen chloride (CK) can be released into indoor air as a liquid spray (aerosol) or as a gas.
- Water: Cyanogen chloride (CK) can be used to contaminate water.
- Food: Cyanogen chloride (CK) can be used to contaminate food.
- Outdoor Air: Cyanogen chloride (CK) can be released into outdoor air as a liquid spray (aerosol) or as a gas.
- Agricultural: If cyanogen chloride (CK) is released into the air as a liquid spray (aerosol), it has the potential to contaminate agricultural products. If cyanogen chloride (CK) is released as a gas, it is highly unlikely to contaminate agricultural products.
Cyanogen chloride (CK) can affect the body by inhalation, ingestion, skin contact, or eye contact.
Personal Protective Equipment
First Responders should use a NIOSH-certified Chemical, Biological, Radiological, Nuclear (CBRN) Self Contained Breathing Apparatus (SCBA) with a Level A protective suit when entering an area with an unknown contaminant or when entering an area where the concentration of the contaminant is unknown. Level A protection should be used until monitoring results confirm the contaminant and the concentration of the contaminant.
NOTE: Safe use of protective clothing and equipment requires specific skills developed through training and experience.
Select when the greatest level of skin, respiratory, and eye protection is required. This is the maximum protection for workers in danger of exposure to unknown chemical hazards or levels above the IDLH or greater than the AEGL-2.
- A NIOSH-certified CBRN full-face-piece SCBA operated in a pressure-demand mode or a pressure-demand supplied air hose respirator with an auxiliary escape bottle.
- A Totally-Encapsulating Chemical Protective (TECP) suit that provides protection against CBRN agents.
- Chemical-resistant gloves (outer).
- Chemical-resistant gloves (inner).
- Chemical-resistant boots with a steel toe and shank.
- Coveralls, long underwear, and a hard hat worn under the TECP suit are optional items.
Select when the highest level of respiratory protection is necessary but a lesser level of skin protection is required. This is the minimum protection for workers in danger of exposure to unknown chemical hazards or levels above the IDLH or greater than AEGL-2. It differs from Level A in that it incorporates a non-encapsulating, splash-protective, chemical-resistant splash suit that provides Level A protection against liquids but is not airtight.
- A NIOSH-certified CBRN full-face-piece SCBA operated in a pressure-demand mode or a pressure-demand supplied air hose respirator with an auxiliary escape bottle.
- A hooded chemical-resistant suit that provides protection against CBRN agents.
- Chemical-resistant gloves (outer).
- Chemical-resistant gloves (inner).
- Chemical-resistant boots with a steel toe and shank.
- Coveralls, long underwear, a hard hat worn under the chemical-resistant suit, and chemical-resistant disposable boot-covers worn over the chemical-resistant suit are optional items.
Select when the contaminant and concentration of the contaminant are known and the respiratory protection criteria factors for using Air Purifying Respirators (APR) or Powered Air Purifying Respirators (PAPR) are met. This level is appropriate when decontaminating patient/victims.
- A NIOSH-certified CBRN tight-fitting APR with a canister-type gas mask or CBRN PAPR for air levels greater than AEGL-2.
- A NIOSH-certified CBRN PAPR with a loose-fitting face-piece, hood, or helmet and a filter or a combination organic vapor, acid gas, and particulate cartridge/filter combination or a continuous flow respirator for air levels greater than AEGL-1.
- A hooded chemical-resistant suit that provides protection against CBRN agents.
- Chemical-resistant gloves (outer).
- Chemical-resistant gloves (inner).
- Chemical-resistant boots with a steel toe and shank.
- Escape mask, face shield, coveralls, long underwear, a hard hat worn under the chemical-resistant suit, and chemical-resistant disposable boot-covers worn over the chemical-resistant suit are optional items.
Select when the contaminant and concentration of the contaminant are known and the concentration is below the appropriate occupational exposure limit or less than AEGL-1 for the stated duration times.
- Limited to coveralls or other work clothes, boots, and gloves.
Emergency Response
- Cyanogen chloride (CK) decomposes on heating, producing toxic and corrosive fumes (hydrogen cyanide, hydrochloric acid, and nitrogen oxides). See the emergency response cards for hydrogen cyanide and hydrochloric acid.
- Cyanogen chloride (CK) reacts slowly with water or water vapor to form toxic hydrogen cyanide and hydrogen chloride. See the emergency response cards for hydrogen cyanide and hydrogen chloride.
- Cyanogen chloride (CK) is incompatible with or may react with most basic and acidic solvents including water.
- Cyanogen chloride (CK) is unstable; it may be stabilized (inhibited) to prevent polymerization.
- Upper and lower explosive (flammable) limits in air are not available for cyanogen chloride (CK).
- Containers may explode when heated or if they are contaminated with water.
- Ruptured cylinders may rocket.
- Fire will produce irritating, corrosive, and/or toxic gases.
- Cyanogen chloride (CK) may burn, but it does not ignite readily.
- For small fires, use dry chemical or carbon dioxide.
- For large fires, use water spray, fog, or regular foam. Move containers from the fire area if it is possible to do so without risk to personnel. Do not get water inside containers. Damaged cylinders should be handled only by specialists.
- For fire involving tanks, fight the fire from maximum distance or use unmanned hose holders or monitor nozzles. Cool containers with flooding quantities of water until well after the fire is out. Do not direct water at the source of the leak or at safety devices; icing may occur. Withdraw immediately in case of rising sound from venting safety devices or discoloration of tanks. Always stay away from tanks engulfed in fire.
- Run-off from fire control may cause pollution.
- If the situation allows, control and properly dispose of run-off (effluent).
- If a tank, rail car, or tank truck is involved in a fire, isolate it for 1.0 mi (1600 m) in all directions; also consider initial evacuation for 1.0 mi (1600 m) in all directions.
- Small spills (involving the release of approximately 52.83 gallons (200 liters) or less)
- First isolate in all directions: 200 ft (60 m).
- Then protect persons downwind during the day: 0.4 mi (0.6 km).
- Then protect persons downwind during the night: 1.8 mi (2.8 km).
- Large spills (involving quantities greater than 52.83 gallons (200 liters)):
- First isolate in all directions: 1400 ft (450 m).
- Then protect persons downwind during the day: 2.7 mi (4.3 km).
- Then protect persons downwind during the night: 6.3 mi (10.1 km).
- Small spills (involving the release of approximately 52.83 gallons (200 liters) or less), when cyanogen chloride is used as a weapon :
- First isolate in all directions: 200 ft (60 m).
- Then protect persons downwind during the day: 0.4 mi (0.7 km).
- Then protect persons downwind during the night: 1.5 mi (2.5 km).
- Large spills (involving quantities greater than 52.83 gallons (200 liters)), when cyanogen chloride is used as a weapon:
- First isolate in all directions: 1300 ft (420 m).
- Then protect persons downwind during the day: 2.5 mi (4.1 km).
- Then protect persons downwind during the night: 5.0 mi (8.1 km).
- Vapors may be heavier than air. They will spread along the ground and collect and stay in poorly-ventilated, low-lying, or confined areas (e.g., sewers, basements, and tanks).
- Hazardous concentrations may develop quickly in enclosed, poorly-ventilated, or low-lying areas. Keep out of these areas. Stay upwind.
- Health: 4
- Flammability: 0
- Reactivity: 2
- Special:

- OSHA: Not established/determined
- NIOSH: Not established/determined
- AIR MATRIX
Deng GC, Zhang ZH, Li B, Gao XD, Zang SL [2001]. Determination of cyanogen chloride in the air pollution by spectrophotometry. Chin J Anal Chem 29(5):565-568.NIOSH [1994]. NMAM Method 6010: Hydrogen cyanide. In: NIOSH manual of analytical methods. 4th ed. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication No. 94-113.NIOSH [1994]. NMAM Method 7904: Cyanides (aerosol and gas). In: NIOSH manual of analytical methods. 4th ed. Cincinnati, OH: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication No. 94-113.
Singh B, Bhise PP, Suryanarayana MVS, Yadav SS, Rao VK, Polke BG, Pandey D, Ganesan K, Rao NBSN [1999]. Silica gel detector tubes for toxic chemicals and their evaluation. J Sci Indus Res 58(1):25-30.
- OTHER
No references were identified for this sampling matrix for this agent. - SOIL MATRIX
EPA [1992]. SW-846 Method 9013: [PDF Format 157 KB] Cyanide extraction procedure for solids and oils. Washington, DC: U.S. Environmental Protection Agency.Matsumura M, Kojima T [2003]. Elution and decomposition of cyanide in soil contaminated with various cyanocompounds. J Hazardous Materials 97(1-3): 99-110. - SURFACES
No references were identified for this sampling matrix for this agent. - WATER
Cancho B, Ventura F, Galceran MT [2000]. Simultaneous determination of cyanogen chloride and cyanogen bromide in treated water at sub-?g/L levels by a new solid-phase microextraction—gas chromatographic—electron-capture detection method. J Chromatogr A 897(1-2):307-315.EPA [1996]. SW-846 Method 9010B: [PDF Format 78 KB] Total and amenable cyanide: distillation. Washington, DC: U.S. Environmental Protection Agency.EPA [1996]. SW-846 Method 9012A: [PDF Format 109 KB] Total and amenable cyanide (automated colorimetric, with off-line distillation). Washington, DC: U.S. Environmental Protection Agency.
EPA [1996]. SW-846 Method 9014: [PDF Format 72 KB] Titrimetric and manual spectrophotometric determinative methods for cyanide. Washington, DC: U.S. Environmental Protection Agency.
Inoue Y, Suzuki Y, Ando M [1993]. Determination of cyanide ion and cyanogen chloride by using photometric detection with 4-pyridinecarboxylic acid-pyrazolone in ion-exclusion. Bunseki Kagaku 42(10): 617-623.
Inoue Y, Suzuki Y, Okubo T [1995]. Determination of cyanide and cyanogen chloride by ion-exclusion chromatography with postcolumn derivatization using an alkaline reduction pretreatment method. Anal Sci 11(5):861-863.
Sclimenti MJ, Hwang CJ, Krasner SW [1996]. A comparison of analytical techniques for the determination of cyanogen chloride in chloraminated drinking-water [Abstracts]. ACS Symposium Series.
Wu WW, Chadik PA, Schmidt CJ [1998]. An in situ synthesis of cyanogen chloride as a safe and economical aqueous standard. Water Res 32(9):2865-2869.
Xie YF, Reckhow Da [1993]. A rapid and simple analytical method for cyanogen chloride and cyanogen-bromide in drinking-water. Water Res 27(3): 507-511.
Signs/Symptoms
Early symptoms of cyanide poisoning include lightheadedness, giddiness, rapid breathing, nausea, vomiting (emesis), feeling of neck constriction and suffocation, confusion, restlessness, and anxiety. Accumulation of fluid in the lungs (pulmonary edema) may complicate severe intoxications. Rapid breathing is soon followed by respiratory depression/respiratory arrest (cessation of breathing). Severe cyanide poisonings progress to stupor, coma, muscle spasms (in which head, neck, and spine are arched backwards), convulsions (seizures), fixed and dilated pupils, and death. The CNS is the most sensitive target organ of cyanide poisoning. Cardiovascular effects require higher cyanide doses than those necessary for CNS effects. In serious poisonings, the skin is cold, clammy, and diaphoretic. Blue discoloration of the skin may be a late finding. Severe signs of oxygen deprivation in the absence of blue discoloration of the skin suggest cyanide poisoning.
Cyanogen chloride (CK) can be rapidly fatal. It severely irritates the eyes, skin, and respiratory tract. It produces whole-body (systemic) effects by interfering with oxygen utilization at the cellular level, with profound central nervous system (CNS), cardiovascular, and respiratory effects. Severe signs of decreased oxygen supply to the tissues (hypoxia) in the absence of bluish discoloration of the skin (cyanosis) are characteristic; cyanosis usually occurs late in the course of poisoning, at the stage of circulatory collapse and cessation of breathing (apnea).
- Intense irritation, severe spasmodic blinking (blepharospasm), and tear production (lacrimation).
- Contact with only the eyes has not been known to result in whole-body (systemic) toxicity, although this is a possibility.
- See Inhalation Exposure.
- Possible bitter, acrid burning taste, followed by constriction or numbness of the throat, salivation, nausea, and vomiting (emesis).
- Whole-body (systemic) toxicity can occur.
- See Inhalation Exposure.
- Mild to moderate: CNS effects: headache, confusion, anxiety, dizziness, weakness (malaise), and loss of consciousness. Cardiovascular effects: palpitations. Respiratory effects: respiratory tract irritation, difficulty breathing or shortness of breath (dyspnea), and transient increase in the rate and depth of breathing (hyperpnea). GI effects: nausea and vomiting (emesis).
- Severe: CNS effects: coma, seizures, and dilated pupils (mydriasis). Cardiovascular effects: shock, abnormal or disordered heart rhythms (dysrhythmias), critically low blood pressure, and cardiac arrest. Respiratory effects: abnormally rapid, followed by abnormally slow respirations; accumulation of fluid in the lungs (pulmonary edema); and respiratory arrest. Eye effects: dilated pupils, inflammation of the surface of the eye, and temporary blindness.
- Irritation.
- Contact with gas or liquefied gas may cause burns, severe injury, and/or frostbite.
- Contact with the skin can contribute to whole-body (systemic) toxicity.
- See Inhalation Exposure.
Decontamination
The purpose of decontamination is to make an individual and/or their equipment safe by physically removing toxic substances quickly and effectively. Care should be taken during decontamination, because absorbed agent can be released from clothing and skin as a gas. Your Incident Commander will provide you with decontaminants specific for the agent released or the agent believed to have been released.
The following are recommendations to protect the first responders from the release area:
- Position the decontamination corridor upwind and uphill of the hot zone. The warm zone should include two decontamination corridors. One decontamination corridor is used to enter the warm zone and the other for exiting the warm zone into the cold zone. The decontamination zone for exiting should be upwind and uphill from the zone used to enter.
- Decontamination area workers should wear appropriate PPE. See the PPE section of this card for detailed information.
- A solution of detergent and water (which should have a pH value of at least 8 but should not exceed a pH value of 10.5) should be available for use in decontamination procedures. Soft brushes should be available to remove contamination from the PPE. Labeled, durable 6-mil polyethylene bags should be available for disposal of contaminated PPE.
The following methods can be used to decontaminate an individual:
- Decontamination of First Responder:
- Begin washing PPE of the first responder using soap and water solution and a soft brush. Always move in a downward motion (from head to toe). Make sure to get into all areas, especially folds in the clothing. Wash and rinse (using cold or warm water) until the contaminant is thoroughly removed.
- Remove PPE by rolling downward (from head to toe) and avoid pulling PPE off over the head. Remove the SCBA after other PPE has been removed.
- Place all PPE in labeled durable 6-mil polyethylene bags.
- Decontamination of Patient/Victim:
- Remove the patient/victim from the contaminated area and into the decontamination corridor.
- Remove all clothing (at least down to their undergarments) and place the clothing in a labeled durable 6-mil polyethylene bag.
- Thoroughly wash and rinse (using cold or warm water) the contaminated skin of the patient/victim using a soap and water solution. Be careful not to break the patient/victim’s skin during the decontamination process, and cover all open wounds.
- Cover the patient/victim to prevent shock and loss of body heat.
- Move the patient/victim to an area where emergency medical treatment can be provided.
First Aid
Careful observation, supplemental oxygen, and supportive care may be sufficient therapy for the patient/victim who does not exhibit physical findings of cyanide toxicity. For the patient/victim exhibiting physical findings of cyanide toxicity, initial treatment consists of administration of antidotes under a physician’s direction, respiratory and circulatory support (oxygen and IV fluids), correction of chemical imbalances in the blood, and seizure control. Speed is critical. Avoid mouth-to-mouth resuscitation regardless of route of exposure. Avoid contact with vomitus, which may off-gas hydrogen cyanide.
Amyl nitrite, sodium nitrite, and sodium thiosulfate are antidotes for cyanide toxicity; however, amyl nitrite and sodium nitrite should not be administered to patient/victims suffering from smoke inhalation. In these cases, only administer sodium thiosulfate. The described administration of nitrites is based on a patient having normal hemoglobin levels. Below normal hemoglobin levels require titration of nitrites.
For mild to moderate physical findings such as nausea, vomiting, palpitations, confusion, anxiety, dizziness (vertigo), and/or abnormally fast or deep respiration (hyperventilation):
- Child (less than 55 lb (25 kg)) Observe the patient/victim and administer 0.75 mL per pound of a 25% solution (1.65 mL per kilogram of a 25% solution) of sodium thiosulfate intravenously over a period of 10 minutes.
- Adult Observe the patient/victim and administer 12.5 g of a 25% solution (50 mL of a 25% solution) of sodium thiosulfate intravenously over a period of 10 minutes.
For severe physical findings such as coma; cessation of breathing (apnea); seizures; slowness of the heart rate, usually to fewer than 60 beats per minute (bradycardia); abnormally low blood pressure (hypotension); bluish skin coloring due to abnormally low levels of oxygen in the blood (cyanosis); irregular heart beat (dysrhythmias); and/or accumulation of fluid in the lungs (pulmonary edema):
- Child (less than 55 lb (25 kg)) Until sodium nitrite becomes available, break one ampule of amyl nitrite into a cloth. Out of every minute, hold the cloth containing amyl nitrite in front of the patient’s mouth for 30 seconds, and then remove it for 30 seconds, until sodium nitrite can be administered. A new ampule of amyl nitrite should be broken into a cloth every 3 minutes. Discontinue use of amyl nitrite when sodium nitrite becomes available. Administration of an entire dose (10 mL of a 3% solution) of sodium nitrite to a child can produce overwhelming lethal methemoglobinemia. Therefore, children should receive 0.15 mL per pound of body weight of sodium nitrite (0.33 mL per kg body weight of 3% sodium nitrite) over a period of 5 to 20 minutes.
- Next, administer 0.75 mL per pound body weight of 25% sodium thiosulfate (1.65 mL per kilogram body weight of 25% sodium thiosulfate) intravenously over a period of 10 minutes. If physical findings persist for 30 minutes after antidote administration, sodium nitrite and sodium thiosulfate may be readministered at half their original respective doses. However, methemoglobin levels should be monitored and not allowed to exceed 40%.
- If a child weighs more than 55 lb (25 kg), administer antidote as described for the adult (see below).
- Adult Until sodium nitrite becomes available, break one ampule of amyl nitrite into a cloth. Out of every minute, hold the cloth containing amyl nitrite in front of the patient’s mouth for 30 seconds, and then remove it for 30 seconds, until sodium nitrite can be administered. A new ampule of amyl nitrite should be broken into a cloth every 3 minutes. Discontinue use of amyl nitrite when sodium nitrite becomes available. Administer 300 mg of a 3% solution (10 mL of a 3% solution) of sodium nitrite intravenously over a period of 5 to 20 minutes.
- Next, administer 12.5 g (50 mL of a 25% solution) of sodium thiosulfate intravenously over a period of 10 minutes. If physical findings persist for 30 minutes after antidote administration, sodium nitrite and sodium thiosulfate may be readministered at half their original respective doses. However, methemoglobin levels should be monitored and not allowed to exceed 40%.
- Immediately remove the patient/victim from the source of exposure.
- Immediately wash eyes with large amounts of tepid water for at least 15 minutes.
- Monitor the patient/victim for signs of whole-body (systemic) effects.
- If signs of whole-body (systemic) poisoning appear, see the Inhalation section for treatment recommendations.
- Seek medical attention immediately.
- Immediately remove the patient/victim from the source of exposure.
- Establish secure large-bore IV access.
- Ensure that the patient/victim has an unobstructed airway.
- Do not induce vomiting (emesis).
- Immediately administer 100% oxygen.
- Prepare a cyanide antidote kit, for use under a physician’s direction, for symptomatic patient/victims. See the Antidote section for antidote administration procedures.
- Treat seizures with benzodiazepines.
- Seek medical attention immediately.
- Immediately remove the patient/victim from the source of exposure.
- Evaluate respiratory function and pulse.
- Ensure that the patient/victim has an unobstructed airway.
- Immediately administer 100% oxygen.
- Assist ventilation as required.
- If breathing has ceased (apnea), provide artificial respiration.
- Establish secure large-bore intravenous (IV) access.
- Prepare a cyanide antidote kit, for use under a physician’s direction, for symptomatic patient/victims. See the Antidote section for antidote administration procedures.
- Monitor for respiratory distress and accumulation of fluid in the lungs (pulmonary edema).
- Seek medical attention immediately.
- Immediately remove the patient/victim from the source of exposure.
- See the Decontamination section for patient/victim decontamination.
- Monitor the patient/victim for signs of whole-body (systemic) effects.
- If signs of whole-body (systemic) poisoning appear, see the Inhalation First Aid section for treatment recommendations.
- Seek medical attention immediately.
Long-Term Implications
Evidence for the benefit of gastric decontamination in cases of cyanide ingestion is limited at best and should come after all other known life-saving measures have been instituted. Gastric lavage (stomach pumping) is recommended only if it can be done shortly after ingestion (generally within 1 hour), in an emergency department, and after the airway has been secured. Activated charcoal may be administered as a slurry (240 mL water/30 g charcoal). Usual dose: 25 to 100 g in adults/adolescents, 25 to 50 g in children (1 to 12 years), and 1 g/kg in infants less than 1 year old. Patient/victims who have ingested cyanogen chloride (CK) or patient/victims who have direct skin or eye contact should be observed in the Emergency Department for at least 4 to 6 hours for the development of delayed symptoms. Patient/victims with significant inhalation exposure should be monitored for the accumulation of fluid in the lungs (pulmonary edema), which occurs more rapidly with exposure to cyanogen chloride (CK) than with exposure to other cyanides.
Usually death occurs rapidly or there is prompt recovery. Survivors of severe cyanide exposures may suffer brain damage due to a direct effect of the poison (toxin) on nerve cells, or to a lack of oxygen, or possibly due to insufficient blood circulation. Examples of long-term neurological effects caused by cyanide poisoning include personality changes, memory loss, and disturbances in movement (both voluntary and involuntary movement disorders); some damage may be permanent.
Information is unavailable about the carcinogenicity, developmental toxicity, or reproductive toxicity from chronic or repeated exposure to cyanogen chloride (CK). Effects of chronic or repeated exposure to cyanogen chloride (CK) are similar to those of cyanide and other cyanide compounds. Chronically exposed workers may complain of headache, eye irritation, easy fatigue, chest discomfort, palpitations, loss of appetite (anorexia), and nosebleeds (epistaxis). Exposure to small amounts of cyanide compounds over long periods of time is reported to cause loss of appetite, headache, weakness, nausea, dizziness, and symptoms of irritation of the upper respiratory tract and eyes.
On-Site Fatalities
- Consult with the Incident Commander regarding the agent dispersed, dissemination method, level of PPE required, location, geographic complications (if any), and the approximate number of remains.
- Coordinate responsibilities and prepare to enter the scene as part of the evaluation team along with the FBI HazMat Technician, local law enforcement evidence technician, and other relevant personnel.
- Begin tracking remains using waterproof tags.
- Wear PPE until all remains are deemed free of contamination.
- Establish a preliminary (holding) morgue.
- Gather evidence, and place it in a clearly labeled impervious container. Hand any evidence over to the FBI.
- Remove and tag personal effects.
- Perform a thorough external evaluation and a preliminary identification check.
- See the Decontamination section for decontamination procedures.
- Decontaminate remains before they are removed from the incident site.
Occupational Exposure Limits
- NIOSH REL:
- Ceiling (15-min): 0.3 ppm (0.6 mg/m3)
- OSHA PEL:
- Not established/determined
- ACGIH TLV:
- Ceiling: 0.3 ppm
- NIOSH IDLH: Not established/determined
- DOE TEEL:
- TEEL-0: 0.75 mg/m3
- TEEL-1: 0.75 mg/m3
- TEEL-2: 1 mg/m3
- TEEL-3: 10 mg/m3
- AIHA ERPG:
- ERPG-1: Not appropriate.
- ERPG-2: 0.4 ppm
- ERPG-3: 4 ppm
Acute Exposure Guidelines
| 5 min | 10 min | 30 min | 1 hr | 4 hr | 8 hr | |
|---|---|---|---|---|---|---|
| AEGL 1 (discomfort, non-disabling) – mg/m3 |
Not established/ determined | Not established/ determined | Not established/ determined | Not established/ determined | Not established/ determined | Not established/ determined |
| AEGL 2 (irreversible or other serious, long-lasting effects or impaired ability to escape) – mg/m3 |
Not established/ determined | Not established/ determined | Not established/ determined | Not established/ determined | Not established/ determined | Not established/ determined |
| AEGL 3 (life-threatening effects or death) – mg/m3 |
Not established/ determined | Not established/ determined | Not established/ determined | Not established/ determined | Not established/ determined | Not established/ determined |
Decontamination (Environment and Equipment)
The following methods can be used to decontaminate the environment/spillage disposal:
- Do not touch or walk through the spilled agent if at all possible. However, if you must, personnel should wear the appropriate PPE during environmental decontamination. See the PPE section of this card for detailed information.
- Keep combustibles (e.g., wood, paper, and oil) away from the spilled agent. Use water spray to reduce vapors or divert vapor cloud drift. Avoid allowing water runoff to contact the spilled agent.
- Do not direct water at the spill or the source of the leak.
- Stop the leak if it is possible to do so without risk to personnel, and turn leaking containers so that gas rather than liquid escapes.
- Prevent entry into waterways, sewers, basements, or confined areas.
- Isolate the area until gas has dispersed.
- Ventilate the area.
Agents can seep into the crevices of equipment making it dangerous to handle. The following methods can be used to decontaminate equipment:
- Not established/determined
Agent Properties
- Chemical Formula:
CNCl - Aqueous solubility:
Soluble - Boiling Point:
57°F (13.8°C) - Density:
Liquid: 1.186 g/mL at 68°F (20°C)
Vapor: 2.16 - Flammability:
Not flammable - Flashpoint:
Not established/determined
- Ionization potential: 12.49 eV
- Log Kbenzene-water:
Not established/determined - Log Kow (estimated):
-0.38 - Melting Point:
21°F (-6°C) - Molecular Mass:
61.47
- Soluble In: All organic solvents.
- Specific Gravity:
1.186 at 68°F (20°C) - Vapor Pressure:
1,010 mm Hg at 68°F (20°C)
1,230 mm Hg at 77°F (25°C) - Volatility:
2,600,000 mg/m3 at 55°F (12.8°C)
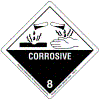
Hazardous Materials Warning Labels/Placards
- Shipping Name:
Cyanogen chloride (stabilized) - Identification Number:
1589 (Guide 125) - Hazardous Class or Division:
2.3 - Subsidiary Hazardous Class or Division:
8 - Label:
Corrosive (UN 1589), Inhalation Hazard; Toxic Gas - Placard Image:

Trade Names and Other Synonyms
- Chlorocyan
- Chlorocyanide
- Chlorocyanogen
In the event of a poison emergency, call the poison center immediately at 1-800-222-1222. If the person who is poisoned cannot wake up, has a hard time breathing, or has convulsions, call 911 emergency services.
For information on who to contact in an emergency, see the CDC website at emergency.cdc.gov or call the CDC public response hotline at (888) 246-2675 (English), (888) 246-2857 (Español), or (866) 874-2646 (TTY).
The user should verify compliance of the cards with the relevant STATE or TERRITORY legislation before use. NIOSH, CDC 2003.